Annals of Neurosciences, Volume 21, Issue 3 (July), 2014
Central depressant and nootropic effects of daytime melatonin in mice
KEY WORDS |
ABSTRACT |
|
Neurobehavior |
Background: Effects of orally administered daytime melatonin on novelty induced behaviors and spatial working memory in mice were evaluated using the open field, the Y maze and the radial arm maze. Purpose: To ascertain the possible nootropic and/or central excitatory or inhibitory effects of daytime oral melatonin in mice. Methods: Adult male mice from our colony, assigned to three and four groups for open field tests and memory tests respectively were given vehicle (normal saline), a standard drug Scopolamine at 0.5 mg/kg i.p, single dose, 30 minutes before behavioral study) or one of two doses of melatonin (5 and 10 mg/kg daily for a period of 30 days). All administrations were done between 8.00 a.m. and 9.00 a.m. daily. Behavioral tests were carried out on day 30 after administration. Results were analysed using a one-way ANOVA followed by a posthoc test (Student-Newman-Keul’s) and expressed as mean ± S.E.M. Results: Open field tests revealed a significant reduction in rearing and grooming behaviors at both doses tested while no significant changes in horizontal locomotion were seen. Y maze studies showed an improvement in spatial memory in mice that received 5 mg/kg of melatonin when compared to scopolamine control. At 10 mg/kg, no significant improvement was seen. A significant increase in the radial arm maze spatial working memory following melatonin administration was seen at 5 mg/kg and 10 mg/kg compared to scopolamine control. Radial arm maze exploration was also significantly reduced. Conclusion: The study demonstrates the ability of exogenously administered melatonin, to affect both central excitation and spatial working memory in mice even when given orally. doi : 10.5214/ans.0972.7531.210304 |
|
Corresponding Author: |
Introduction
Melatonin is a neurohormone secreted by the pineal gland. Its secretion follows strictly a circadian rhythm with the acrophase occurring during the dark phase and the nadir during the light phase in both nocturnal and diurnal animals.1,2 Its amino acid precursor is tryptophan which undergoes a stepwise conversion, first to hydroxytryptophan and then to hydroxy tryptamine and eventually to melatonin. Although melatonin is primarily synthesized within the pineal gland, it can be found in the retina and some other sites. In mammals however, a large proportion of the hormone, if not all, found peripherally is believed to be derived from the pineal gland, being that pinealectomy abolishes its synthesis and function.3 Pinealectomy also abolishes the rhythm of melatonin secretion.
Melatonin was first isolated and named in 1958 by Lerner and his colleagues.4 McCord and Allen in 1917 first discovered that feeding extracts of cow pineal gland to tadpoles caused lightening of their skin due to contraction of the dark epidermal melanophores, this is believed to be related to the mechanism by which some amphibians and reptiles change the color of their skin.5 Studies have also focused on the possible role of melatonin on pigmentation in lower vertebrates and gonadal maturation in mammals.6 Melatonin is present in plants and some food crops like rice, corn, tomato and other edible fruits.7,8 It modulates a number of physiological functions e.g. sleep, circadian, visual, cerebrovascular, reproductive, neuroendocrine, and neuroimmunological functions.9 Coupled with its effects on body physiology it also influences behavioral processes like learning and depression.10 Melatonin receptor binding sites have been found in brain regions associated learning and memory.11 Studies have shown that melatonin reduces recognition time resulting in the facilitation of short-term memory12 together with its effects on passive and active avoidance learning;13 it can be safe to say that melatonin may be beneficial in enhancing learning and memory. Most studies involving melatonin have concentrated on intravenous, intraperitoneal,14–17 intra-amygdala,4 and intra-cerebroventricular routes of administration and those that have used the oral route have studied the effects of melatonin when added to drinking water and not as a bolus as used in our study. Secondly, the duration of most melatonin studies have been for the most part 14 days with most researchers studying acute effects of melatonin.18 We wanted to examine possible effects of daily oral administration of timed release melatonin when used over a longer duration of time.
Methods
Reagents and drugs
Normal Saline, Scopolamine hydrobromide and Melatonin Time Release tablets (NATROL Inc. USA) which were crushed into fine powder, weighed and dissolved in measured volume of isotonic saline solution to get desired concentrations. Melatonin at two doses (5 and 10 mg/kg) was administered orally using a cannula.
Animals
Adult male Swiss albino mice (Empire Breeders, Osogbo, Nigeria) weighing 20 to 25 g were used. Mice were housed in plastic cages measuring 16 × 12 × 10 inches (6 mice in each cage). Housing is a temperature-controlled (22.5°C ± 2.5°C) quarters with 12 hours of light. Mice had free access to food and water except during the behavioral tests. The experimental protocol was approved by the University Animal Ethics Committee. All rules applying to animal safety and care were observed.
Study design
The behavioral models used were the open field, the Y maze and the radial arm maze. Mice in respective groups received vehicle (normal saline), standard drug (Scopolamine 0.5 mg/kg i.p. single dose) or one of two doses of melatonin (5 and 10 mg/kg respectively for a period of 30 days). Tests were carried out after the last dose of melatonin. The behavioral tests were conducted in a large quiet room between the hours of 8 a.m. and 9 a.m. At the beginning of the test, each animal was placed in the apparatus and its behavior was videotaped for subsequent analysis.
Behavioral testing
Locomotion, rearing and grooming was observed and scored at 10 minutes interval. This was used to characterize behavioral changes in the mice, when placed in the open field. The open field is a rectangular area composed of floor measuring 36 × 36 × 26 cm. The floor was divided by permanent red markings into 16 equal squares at the bottom. Generally, spontaneous motor activity was monitored for 30 minutes in the open field as described by Ajayi and Ukponmwan.19 After treatment, as earlier explained, each mouse was introduced into the field and the total locomotion (number of floor units entered with all paws), rearing frequency (number of times the animal stood on its hind legs or with its fore arms against the walls of the observation cage or free in the air) and frequency of grooming (number of body cleaning with paws, picking of the body and pubis with mouth and face washing actions) within each 10 minute interval were recorded.
The Y and radial arm mazes were used to measure general activity and spatial working memory. Spontaneous alternation was assessed using a Y- maze composed of three equally spaced arms (120°, 41 cm long and 15 cm high). Each mouse was placed in one of the arm compartments and was allowed to move freely until its tail completely entered another arm. An alternation is defined as entry into all three arms consecutively, the number of maximum spontaneous alternations is the total number of arms entered minus two, and the percentage alternation is calculated as {(actual alternations/maximum alternations) × 100}. For each animal the Y-maze testing was carried out for 5 minutes.20 Working memory in the radial arm maze was measured as sequential arm entries before error. The apparatus consists of eight equidistantly spaced arms, each about 4 feet long, and all radiating from a small circular central platform. Working memory is assessed when the mouse enters each arm a single time. Re-entry into the arms would result in a working memory error.21 General activity in both mazes was measured as the total number of arm entries.
Statistical Analysis
Results are expressed as mean ± S.E.M. Descriptive and inferential statistics was applied to the data using Graphpad prism 5 analysis software (Graphpad software Inc., San Diego California, USA), p values less than 0.05 were considered to be statistically significant.
Results
Effect of melatonin on arm entry before first error
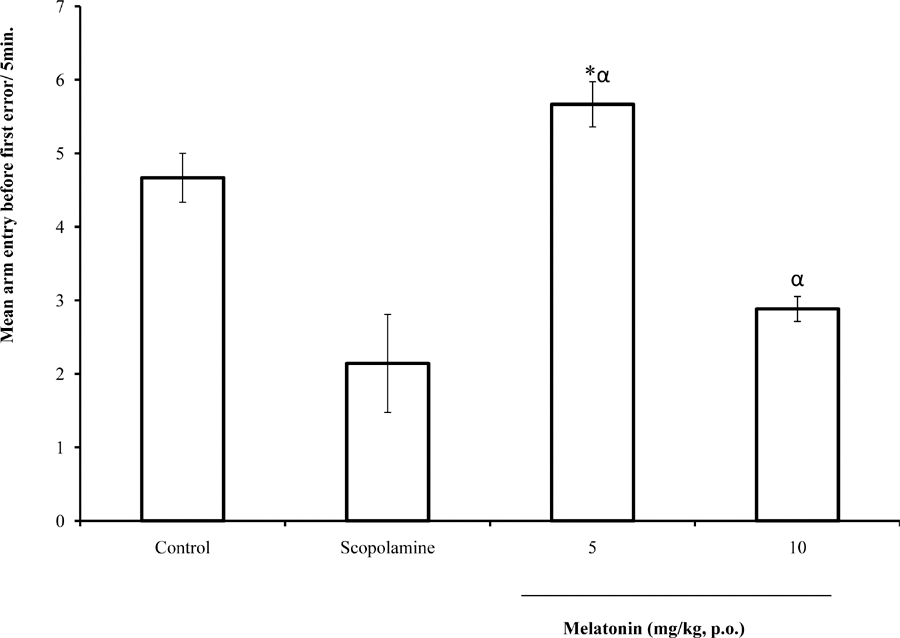
Following 5 minutes of exposure to the radial arm maze, there was a significant (F = 15.27, p<0.05) increase in spatial memory score (arm entry before first error) at 5 and 10 mg/kg of melatonin compared to Scopolamine control; when compared to saline control, there was a reduction in arm entry before first error at 10 mg/kg whereas the difference seen at was not significant Figure 1.

Fig. 1: Effect of melatonin on arm entry before first error in the radial arm maze. Each bar represents Mean ± S.E.M, *αp≤0.05 compared to the control, n = 7.
Effect of melatonin on total arm entry
Following 5 minutes of exposure to the radial arm maze, there was a significant {(F = 125.25, p<0.05) reduction in total arm entry at 10 mg/kg of melatonin compared to saline control, however at 5 mg/kg the difference was not significant (Figure 2).
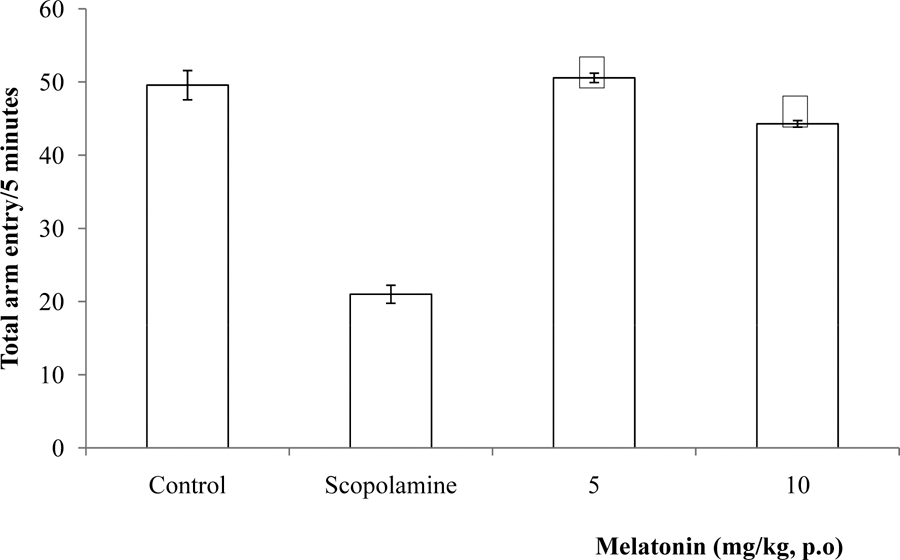
Fig. 2: Effect of melatonin on total arm entry in the radial arm maze. Each bar represents Mean ± S.E.M, *p≤0.05 compared to the control, n = 7.
Effect of melatonin on % alternation in the Y maze
Following 5 minutes of exposure to the Y maze, there was a significant (F = 46.04, p<0.05) increase in spatial memory scores (% correct alternation) at 5 mg/kg of melatonin compared to Scopolamine control at 10 mg/kg the difference was not significant; when compared to saline control there was a reduction in proportional correct alternation at 5 and 10 mg/kg (Figure 3).
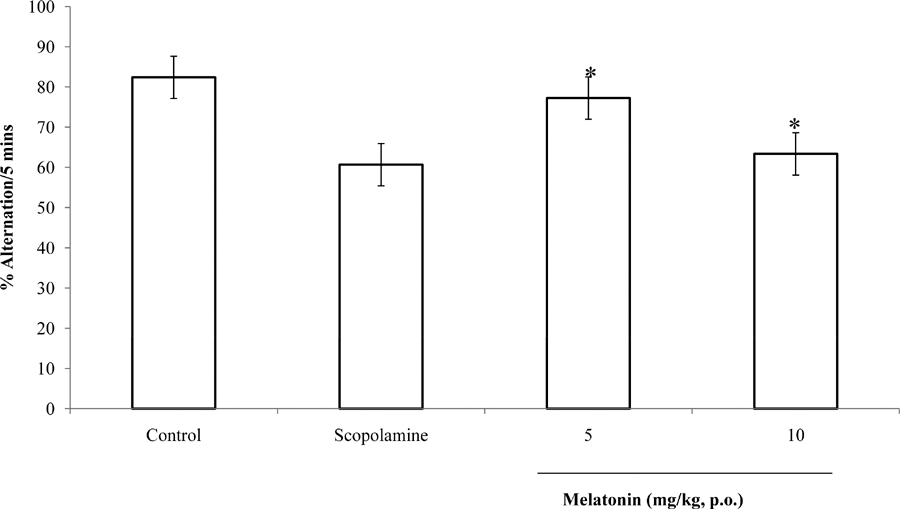
Fig. 3: Effect of melatonin on % alternation in the Y maze. Each bar represents Mean ± S.E.M, *p≤0.05 compared to the control, n = 7.
Effect of melatonin on horizontal locomotion
Following 30 minutes of exposure to the open field, there was no significant difference in locomotor activity following administration of melatonin at either dose compared to saline control (Figure 4).
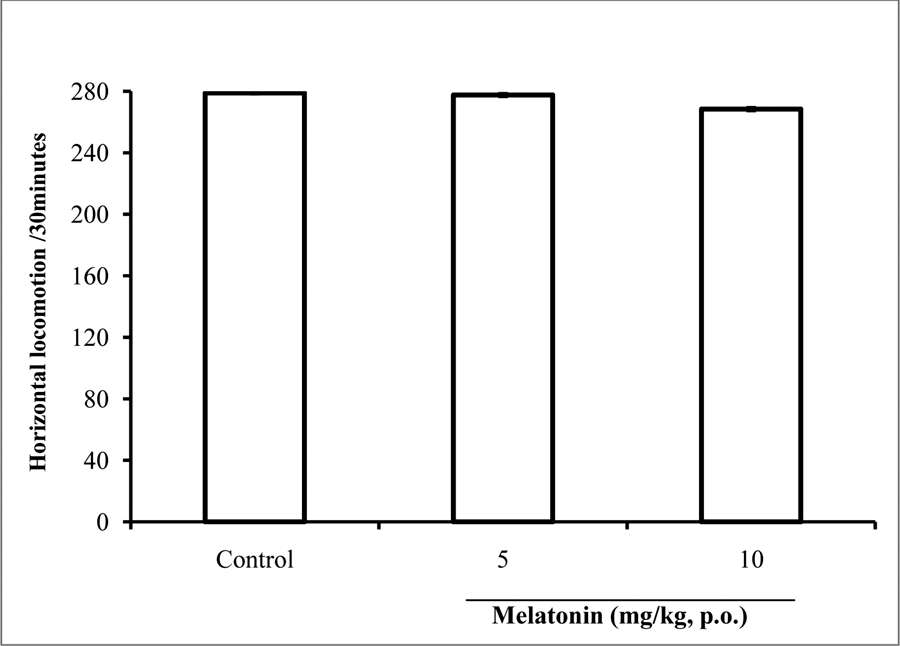
Fig. 4: Effect of melatonin (5 and 10 mg/kg) on locomotor activity in the open field box. Each bar represents Mean ± S.E.M, *p≤0.05 compared to the control, n = 7.
Effect of melatonin on rearing activity
Following 30 minutes of exposure to the open field, there was a significant {(F = 166.00, p<0.05) reduction in rearing activity at both doses of melatonin compared to control (Figure 5).
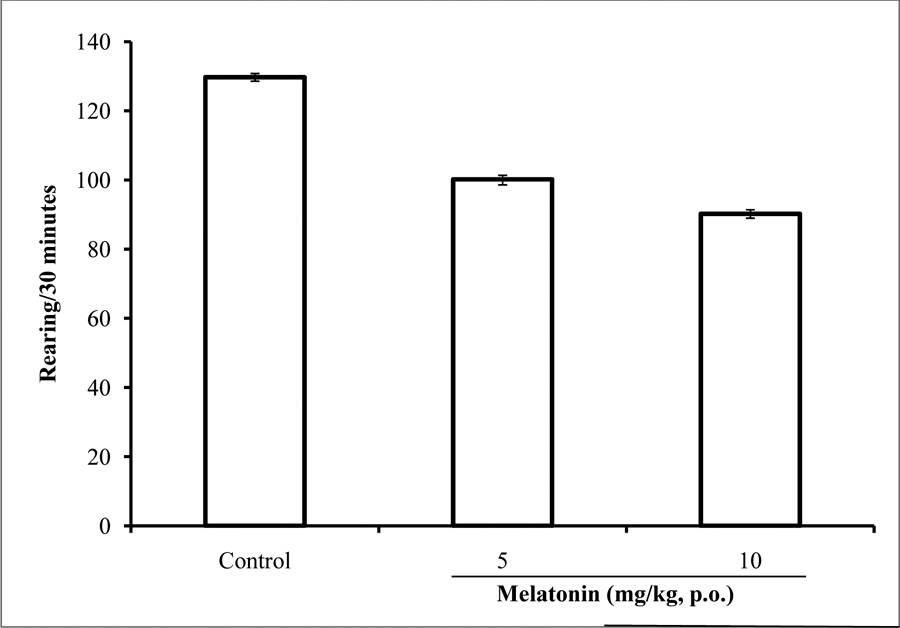
Fig. 5: Effect of melatonin (5 and 10 mg/kg) on rearing activity in the open field box. Each bar represents Mean ± S.E.M, *p≤0.05 compared to the control, n = 7.
Effect of melatonin on grooming behaviour
Following 30 minutes of exposure to the open field there was a significant (F = 12.80, p<0.05), reduction in grooming behaviour in the groups of animals that received melatonin compared to controls (Figure 6).
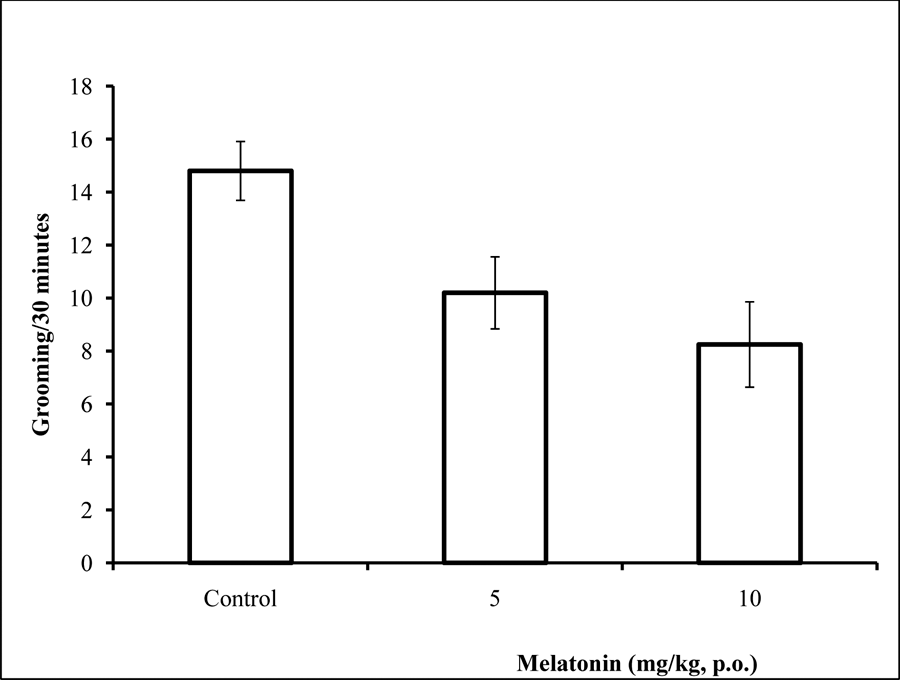
Fig. 6: Effect of melatonin (5 and 10 mg/kg) on grooming behavior in the open field box. Each bar represents Mean ± S.E.M, *p≤0.05 compared to the control, n = 7.
Discussion
An assessment of the effects of oral melatonin (TR) on novelty induced behaviors and working memory in mice was made. Melatonin modulates a number of physiological functions, some of which include sleep, immune functions, contraction of smooth muscle and body temperature.22 Its behavioral effects were examined after thirty days of a daily dosing regimen using three behavioral models.
The present study revealed that in the radial arm maze, sub chronic administration of oral timed release formulations of melatonin resulted in a significant increase in working memory compared to the scopolamine induced of memory loss. In comparison to the saline controls, melatonin administered at 10 mg/kg actually led to a decrease in performance. Melatonin also caused a significant reduction in total arm entry at 10 mg/kg whereas at 5 mg/kg the difference was not significant.
The Y-maze is a behavioral model that can be used to examine locomotor activity as well as learning and memory; it assesses hippocampus- dependent navigational behaviors of rodents.23 The results of the Y maze studies showed an improvement in spatial memory scores in mice that received 5 mg/kg of melatonin when compared to scopolamine control, at 10 mg/kg, no significant improvement was seen.
The neurotransmitter acetylcholine (ACh) is known to be important in learning and memory processes in the hippocampus as evident by association of cholinergic activity with the hippocampus is related with memory. Subnormal activity is associated with spatial memory impairment as shown with drugs that impair cholinergic transmission e.g. scopolamine as used in this study. The radial arm maze is an appetitive motivated task useful in assessing spatial reference as well as spatial working memory performance and factors affecting these processes.24 The radial arm maze is undoubtedly one of the most commonly used maze for assessing working memory in rodent studies,21 the two commonly used measures of performance are the number of arms in the maze and memory capacity.
Working memory can be defined as short term memory of an object, stimulus, or location used within a testing session, but not necessarily between sessions, this differs from reference memory, which is memory that would typically be acquired following repeated training, and persist long periods of time usually days to months. In the radial arm maze, melatonin caused an improvement in spatial working memory at both doses used when compared to scopolamine control. The effects of melatonin on spatial learning and memory processes have been documented and differ in many respects although quite a number of studies have reported that melatonin given via the oral, intraperitoneal or any route in rodents resulted in improvement in learning and memory processes especially in models of memory loss25,26 and advocacy on its suitability in the management of Alzheimer’s disease and mild cognitive impairment is ongoing. On the contrary however, some studies have also demonstrated its ability to impair cognition in healthy humans, as evidenced by a deterioration in performance in response and reaction time scores in visual and auditory tasks27 also, impaired performance in a psychomotor digit-symbol substitution test have also been reported.28 Soto Moyano et al29 reported a decline in the visuospatial performance of rats that received exogenous melatonin 30 minutes before radial arm tests.
Some earlier studies had suggested that hypothermic properties of melatonin may result in a reduction of cerebral processing speed,30 affecting cognition indirectly rather than by a direct effect on brain areas that are relevant to cognition. While this theory of ’cerebral freezing’ is rather fascinating to us, we believe that effects of melatonin on memory can be demystified by a meticulous search for an understanding of how it may influence neurotransmitters that are involved in memory processes. The present study shows that melatonin improves memory significantly in comparison to a model of memory loss; while on the contrary, it actually caused a reduction in memory task performance in healthy mice. We believe that melatonin’s effect on memory processes is probably driven through an enhancement of hippocampal cholinergic transmission when there is a pre-existing deficit, however, the influence is not strong enough to fully restore memory functions.
How melatonin exerts these effects on neurobehavior is still subject to continuous study, it has, however, been suggested that the neurobehavioral effects of melatonin use could be secondary to reduced levels of psychomotor vigilance, as revealed by studies that demonstrated increased reaction time scores while evaluating alertness during sustained attention.31
The results of the open field test showed a reduction in rearing and grooming behavior while locomotor activity was not significantly different from saline control. The open field is a commonly used behavioral test with horizontal and vertical locomotion being commonly recorded behaviors.32 Both locomotor and rearing activities of rodents are central excitatory behaviors and indicative of the rodent’s explorative ability.19 A reduction in rearing in rodents point to a central inhibitory effect. The results of this study are consistent with existing literature in that studies have demonstrated that administration of melatonin produced differential behavioral responses that were strongly dependent on experimental design. Glombleck et al reported a reduction in horizontal locomotion in adult golden hamsters after intraperitoneal injection of 30 microgram/kg of melatonin daily for five days.16 In another study this time by Kopp and collaborators administering 10 mg/kg of melatonin to adult C3H/He mice revealed a significant reduction in horizontal locomotor and rearing;33 the present study demonstrated a reduction in rearing activity at both 5 and 10 mg/kg of melatonin administered daily over a 30 day period while locomotor activity did not show any significant difference from control, our results however differ from that of Ripple34 whose work on the effects of prenatal exogenous melatonin administration resulted in an increase in locomotor activity and rearing which was blunted by the administration of luzindole; a melatonin receptor antagonist. A reduction in central excitatory behaviors as seen here is in line with a reduced excitatory input such as glutamatergic or an increased inhibitory input such GABAergic or both. Our assumption is that melatonin acts at least by enhancing cortical GABAergic input.
Grooming is a very important and ancient behavior that can be observed in a number of animals.35 It serves a wide range of purposes extending beyond the primary goal of hygiene and body care, to include thermoregulation, chemo-communication, social interaction and stress reduction.36 Grooming is modulated from a number of brain regions, and by neuromediators, hormones and drugs.37,38 In this study administration of melatonin resulted in a suppression of grooming behavior. It is known that pelage cleaning in rodents increases with exposure to novelty39 and this response is modulated by, central dopaminergic activation via D1 receptors40 although inputs from the GABA system has also been implicated in the expression of novelty- induced grooming via its GABA A and GABA B receptors38 this it does by reducing grooming behavior so drugs that inhibit GABA may cause an increase in grooming and vice versa. Melatonin administration over a 12 week period was also reported to have reduced grooming and other mating behaviors in voles41 and rabbits42 and hamsters.43,44
Conclusion
Exogenously administered oral melatonin is able to affect both central excitation and spatial working memory in mice.
This article complies with International Committee of Medical Journal Editor’s uniform requirements for manuscript.
Conflict of interest: None.
Competing interests: None, Source of funding: None
Received Date : 20 April 2014; Revised Date : 17 May 2014;
Accepted Date : 20 June 2014
References
1. Comai S, Gobbi G. Unveiling the role of melatonin MT2 receptors in sleep, anxiety and other neuropsychiatric diseases: novel target in psychopharmacology. J Psychiatry Neurosci 2014; 39 (1): 6–21.
2. Arendt J. Melatonin. Clin Endocrinol (Oxf) 1988; 29: 205–29.
3. Hoffman RA. And Reiter RJ. Rapid pinealectomy in hamsters and other small rodents. Anat Record 1965; 24: 83–9.
4. Alper Karakas and Hamit Coskun: Intraamygdalar melatonin administration and pinealectomy affect anxiety like behavior and spatial memory. Neuroendocrinology and Behavior, INTECH 2012; chapter 2 http://dx.doi.org/10.5772/4869.
5. Sugden D, Davidson K, Hough KA et al. Melatonin, melatonin receptors and melanophores: a moving story. Pigment Cell Res. 2004; 17(5): 454–60.
6. Filadelfi AM, Castrucci AM. Comparative aspects of the pineal/melatonin system of poikilothermic vertebrates. J. Pineal Res. 1996; 20(4): 175–86.
7. Tan DX, Hardeland R, Manchester LC, et al. Functional roles of melatonin in plants, and perspectives in nutritional and agricultural science. J. Exp. Bot. 201; 63(2): 577–97.
8. Lamont KT, Somers S, Lacerda L et al. Is red wine a SAFE sip away from cardioprotection? Mechanisms involved in resveratrol- and melatonin-induced cardioprotection. J. Pineal Res. 2011; 50(4): 374–80.
9. Arendt, J. Melatonin, circadian rhythms and sleep. New England J Med 2000; 343: 1114–1116.
10. Loiseau F, Bihan CL. Hamon M, et al. Effects of melatonin and agomelatine in anxiety-related procedures in rats: Interaction with diazepam. Eur Neuropsychopharm 2006; 16: 417–428.
11. Weaver DR, Rivkees SA, Reppert SM. Localization and characterization of melatonin receptors in rodent brain by in vitro aotoradiography. J Neurosci 1989; 9: 2581–2590.
12. Argyriou A, Prast H, Philippu A, et al. Melatonin facilitates short-term memory. Eur J Pharmacol 1998; 349:159–162.
13. Kovács GL, Gajari I, Gyula T, et al. Effects of melatonin and pinealectomy on avoidance and exploratory activity in the rat. Physiol Behav 1974; 13: 349–55.
14. Farrin BB , Samad Z. Melatonin improves spatial navigation memory in male diabetic rats Vet. Res. Forum. 2012; 3(3): 187–192.
15. Kalonia H, Kumar A. Protective effect of melatonin on certain behavioral and biochemical alterations induced by sleep-deprivation in mice. Indian J Pharmacol 2007; 39: 48–51.
16. Golombeck DA, Escolar E, Cardinali DP. Melatonin-induced depression of locomotor activity in hamsters: Time-dependency and inhibition by the central type benzodiazepine antagonist Ro 15-1788. Physiol and Behav. 1991; 49: 1091–1097.
17. Stetson MN, Tay DE. Time course of sensitivity of golden hamsters to melatonininjections throughout the day. Biol Reprod 1983; 29: 432–38.
18. Corina V, Bianca B, Crişan D et al. Neuroprotective Effect Of Melatonin In Experimentally Induced Hypobaric Hypoxia Rom. J Morphol Embryol 2013; 54(4): 1097–1106.
19. Ajayi AA, Ukponmwan OE. Evidence of Angiotensin II and Endogenous opioid Modulation of NIR in the Rat. Afr J of Med & Med Sci, 1994; 23: 287–290.
20. Olakunle O, Adejoke O. Subchronic oral bromocriptine methanesulfonate enhances open field novelty induced behaviours and spatial memory in male Swiss albino mice. Neuroscience journal; 2012; 2013 Article id 948241.
21. Olton DS, Samuelson RJ. Remembrance of places passed: spatial memory in rats. J Exp Psychol: Anim Behav Process. 1976; 2: 97–116.
22. Macchi MM, Bruce JN. Human pineal physiology and functional significance of melatonin. Front Neuroendocrinol 2004; 25: 177–195.
23. Conrad CD, Grote KA, Hobbs RJ, et al. Sex differences in spatial and non-spatial Y-maze performance after chronic stress. Neurobio Learn Mem. 2003; 79: 32–40.
24. Kulkarni SK. “Handbook of experimental pharmacology” 3rded, Vallabh Prakashan Delhi, 2005; 56–66.
25. Andrea C, Paula M, García S, et al. Long-term oral administration of melatonin improves spatial learning and memory and protects against cholinergic degeneration in middle-aged Ts65Dn mice, a model of Down syndrome. J of Pineal Res 2013; 54(3): 346–358.
26. Olcese JM, Cao C, Mori T, et al. Protection against cognitive deficits and markers of neurodegeneration by long-term oral administration of melatonin in a transgenic model of Alzheimer disease. J Pineal Res. 2009; 47(1): 82–96.
27. Rogers NL, Phan O, Kennaway DJ, et al. Effect of daytime oral melatonin administration on neurobehavioral performance in humans. J Pineal Res 1998; 25: 47–53.
28. Naguib M, Samarkandi AH. Premedication with melatonin: a double-blind, placebo-controlled comparison with midazolam. Br JAnaesth 1999; 82: 875–80.
29. Soto-Moyano R, Burgos H, Flores F, et al. Melatonin administration impairs visuo-spatial performance and inhibits neocortical long-term potentiation in rats. Pharmacol Biochem Behav. 2006; 85(2): 408–14.
30. Slotten HA, Krekling S. Does melatonin have an effect on cognitive performance? Psychoneuroendocrinol 1996; 21: 673–80.
31. Graw P, Werth E, Krauchi K, et al. Early morning melatonin administration impairs psychomotor vigilance. Behav Brain Res 2001; 121: 167–72
32. Crusio WE. Genetic Dissection of Mouse Exploratory Behaviour. Behav Brain Res 2001; 125(1–2): 127–132.
33. Kopp C, Vogel E, Rettori MC, et al. The effects of melatonin on the behavioral disturbances induced by chronic mild stress in C3H/HE mice. Behav. Pharmacol. 1999; 10: 73–83.
34. Ripple J. The Effects of Altered Prenatal Melatonin Signaling on Adult Behavior and Hippocampal Gene Expression of the Male Rat: A Circadioneuroendocrine-Axis Hypothesis of Psychopathology. 2010; Paper 30. http://digitalcommons.bucknell.edu/honors_theses/30.
35. Sachs BD. The development of grooming and its expression in adult animals. Ann N Y Acad Sci 1988; 525: 1–17.
36. Terry RL. Primate grooming as a tension reduction mechanism. J Psychol, 1970; 76: 129–36.
37. Spruijt BM, van Hooff JA, Gispen WH, et al. Ethology and neurobiology of grooming behavior. Physiol Rev 1992; 72: 825–52.
38. Barros HM, Tannhauser SL, Tannhauser MA, et al. The effects of GABAergic drugs on grooming behaviour in the open field. Pharmacol Toxicol 1994; 74: 339–44.
39. Stoessl AJ. Dopamine D1 receptor agonist induced grooming is blocked by the opiod receptor antagonist maloxone. Eur. J. of Pharmacolo. 1996; 259: 301–303.
40. Komorowska J, Pellis S M. Regulatory mechanisms underlying novelty-induced grooming in the laboratory rat. Behav. processes, 2004; 67(2): 287–293.
41. Ferkin MH, Kile JR. Melatonin treatment affects the attractiveness of the anogenital area scent in meadow voles (Microtus pennsylvanicus). Horm Behav. 1996; 30(3): 227–35.
42. Goldman BD and Nelson RJ (1993) Melatonin and seasonality in mammals. In Melatonin: Biosynthesis, Physiological Effects, and Clinical Applications, HS Yu and RJ Reiter, eds, pp 225–252, CRC Press, Boca Raton, FL.
43. Fleming AS, Phillips A, Rydall A, et al. Effects of photoperiod, the pineal gland and the gonads on agonistic behavior in female golden hamsters (Mesocricetus auratus). Physiol Behav. 1988;44: 227–234.
44. Karp JD, Dixon ME, Powers JB. Photoperiod history, melatonin, and reproductive responses of male Syrian hamsters. J Pineal Res. 1990; 8: 137–152.