Annals of Neurosciences, Vol 13, No 1 (2006)
Annals of Neurosciences, Volume 13, Issue 1 (January), 2006
ALUMUNIUM MEDIATED OXIDATIVE STRESS: POSSIBLE RELATIONSHIP TO COGNITIVE IMPAIRMENT OF ALZHEIMERS TYPE
Corresponding author
Abbas Ali Mahdi
Departments of Biochemistry, King George's Medical University, Lucknow 226003
Abstract
Aluminum (Al) is a trivalent cation that does not undergo redox change and it may cause a variety of neurological disorders. In this review, we discuss evidence for a significant role of aluminum in Alzheimer's disease (AD). Al has been implicated as a potential risk factor in AD and for elderly cognitive impairment by epidemiological studies of drinking water, food, medicine as well as natural resources. Neurotoxicity from excess brain exposure to aluminum is documented from both clinical observations and animal experiments. The pathology of Alzheimer's disease is related to the alteration in neurotransmission, beta amyloid production, plaques formation and cytoskeletal abnormalities. The exact mechanism of Al toxicity is still unclear; however accumulating evidences suggested that Al could potentiate oxidative and inflammatory events leading to tissue damage. In conclusion it is proposed that a combination of anticholinergic drugs and micronutrients including antioxidants may be more effective than the individual agents in prevention and treatment of Alzheimer's disease.
Introduction
Aluminum is the most prevalent metal and the third most important abundant element in earth's crust, only oxygen (49.5%) and silicon (26%) occur more commonly than aluminum (8%). In biological systems, aluminum is present only in trace amounts. In no case, Al 3+ has been shown to have a definite biological function and it is poorly absorbed and efficiently eliminated when absorption does occur; Al is described mainly in bone, liver, testes, kidney, and brain (1). The salts of aluminum are soluble in water and are, therefore, easily assimilated in to the body and may be deposited in to the brain and neuronal tissue and accumulate there.
The subject of aluminum toxicity has been controversial for the last around three decades and its toxicity (depending on amounts) has been known over a hundred years. Epidemiology, autopsy, therapy and laboratory studies provide rich direct evidence that aluminum is one of the primary risk factors for the development of Alzheimer's disease and Al is also an indirect factor of AD. Aluminum is found in our food supply, in the air and soil, and is bound to bauxite in nature. Drinking water has been identified as a potential route of Aluminum exposure and concomitant neurotoxicity in both laboratory animals and humans (2).
Al was first recognized as human neurotoxin in 1886, in Prussian army studies of amputees whose wounds were treated with Al to staunch bleeding (3). The first laboratory animal study linking Al to brain damage was published in 1973 (4). The Al hypothesis was born in 1965 when Wisniewski, Terry and Klatzo showed that introduction of Al salt into the brains of rabbits induces the formation of neurofibrillary tangles that appeared similar to the neurofibrillary tangles of Alzheimer's disease (5–7). Berlyne and co workers (8) reported increased sensitivity to the toxic effect of Al in rats with impaired renal function and cautioned against the use of Al salts in humans with renal failure. Aluminum as a possible cytotoxic factor in the mechanism associated with neurofibrillay degeneration in senile and presenile dementia of Alzheimer type was suggested by Crapper and associates (9). The hypothesis that this form of Al toxicity might be important in the process associated with neurofibrillary degeneration in human disease, including senile and presenile dementia of the Alzheimer type, was investigated by several laboratories (10–12).
Aluminum hypothesis and Alzheimer's disease
Three routes have been proposed by which Al could enter the brain from systematic circulation; blood brain barrier (BBB), nasal olfactory pathway and cerebrospinal fluid (CSF) (13,14). More rapid exchange is possible through the BBB, via transferrin (TF) mediated transport of Al (15). Another important carrier for brain Al influx may be monocarboxylate transporter (MCT), a proton co-transporter, which is located at both the luminal and abluminal surfaces of the BBB (16). As mentioned earlier aluminum hypothesis began when Wisniewski, Terry and Klatzol's demonstrated AD like neurofibrillary pathology by injection of Al salts into the rabbit brain. However, subsequent work by these investigators showed that the similarities between Al involved tangles and those of AD were more apparent than real. As reviewed in Wisniewski and Wen (17) Al induced tangles and AD pathology appeared similar only if the tissue was treated with silver staining and then viewed under the light microscope. Analysis showed that AD tangles are in effect quite different from the pathology induced by Al. Aluminum induced tangles differ from those of AD in their distribution at both gross and ultra structural levels (Table no. 1).
| Tangle Characteristics | Aluminum-Induced | AD |
|---|---|---|
| Protein composition | Neurofilament | Tau |
| Configuration | Single, Straight Filaments | Paired Helical Filaments |
| Diameter | 10 nM | 20–24 nM |
| Building block | 2.0 nM Protofilaments | .2 nM Protofilaments |
| Intraneuronal localization | Cell body, Proximal Protein of Dendrites and Axons |
Entire Neuron |
| Regional Localization | Forebrain, Spinal Cord | Forebrain |
Epidemiological studies
Several studies indicate that chronic Al exposure may have long-term detrimental effects. All elderly persons face Al exposure putting them at potential risk of injury to health and brain function. Only small proportion of elderly people progress to cognitive impairment or dementia, though this proportion doubles every five years after a population reaches 65 years of age (18).
Alzheimer's disease is the most common form of adult onset dementia (19). It is fourth leading cause of death in western countries, preceded only by heart disease, cancer and stroke (20). Currently, there are estimated two million AD patients in India and 17–25 million worldwide (21). Age is by far the main risk factor for AD. Its prevalence illustrates an exponential risk with age, approximately doubling each five year period, from 1% incidence at the age 65 years to 16–20% at 85 years (18). Ecological studies have suggested that concentration of aluminum in drinking water of 0.10–0.20 mg/l may increase the risk of Alzheimer's disease with relative risks ranging from 1.35–2.6 (4, 22–25). All the epidemiological studies, however, have ignored the greatest source of Al for the ordinary citizen, viz foods. According to World Health Organization (WHO), the provisional food weekly intake (PTWI), Al as a contaminant (also as an additive) is 7 mg/kg daily weight for adults, for children the acceptable daily intake (ADI) is 2 mg.
Aluminum mediated pathological lesions in brain
Alzheimer's disease is a progressive dementia, characterized clinically by a specific pattern of memory impairment, followed by cognitive deficits. The confirmed diagnosis of the disease is made histologically by the presence of numerous amyloid containing senile plaques and neurofibrillary tangles in Alzheimer's brain, it is possibly correlated to the memory impairment of the patients, whereas hypophosphorylated tau is the major component of the tangles. Al is a recognized neurotoxin that is reported to be one of the possible factors responsible for AD and it may be the source of initial injury. This metal is a known toxin to the nervous system to cause brain cell death, senile plaques and neurofibrillary tangles (26). It has been reported that Al3+ accumulated in the human brain at 6 microgram per year of life as well as an autopsy composition of the brain of normal elderly people aged 75 to 101 with those of younger persons aged 32 to 46 measured a dramatic increase of bulk aluminum levels (27). Hippocampus aluminum content averaged 28 times higher in the non-demented elderly than in the younger people and its level in frontal cortex averaged 19 times higher. The brain of these elderly people showed hallmark changes typical of AD, such as senile plaques and neurofibrillary tangles. The patient with Alzheimer's disease is neurobiologically characterized by the occurrence of a minimum density of neurofibrillary tangles and neurotics plaques in the hippocampus and the associated cortex of brain.
Armstrong et al. (28) reported that aluminum enters the individual neurons where it may be involved in the production of abnormal tau protein leading to the formation of cellular neurofibrillary tangles, neuropil threads and the processing of amyloid precursor protein resulting in the formation of beta amyloid deposits and senile plaques. Tau protein is the major component of paired helical filaments (PHFs), which form a compact filamentous network described as neurofibrillary tangles (NFTs). Although neurofibrillary tangles are more closely associated with neuronal death than beta amyloid, the evidence is becoming convincing that beta amyloid is the factor most responsible for starting the degenerative processes of Alzheimer's disease. Neurofibrillary tangles are bundles of filaments inside the neuron that abnormally twist around one another. Numerous neurofibrillary tangles are found in area of the brain associated with memory and learning (hippocampus), fear and aggression (amygdala), and thinking (cerebral cortex). It is believed that neurofibrillary tangles play a role in the memory loss and personality changes associated with AD.
The beta amyloid peptide present in the core of senile plaques is a 42 amino acid chain produced by cleavage of a longer protein known as amyloid precursor protein (APP). It has been reported that APP is normally found embedded in neural membrane where it may serve to stabilize the contact points between synapses (29). Furthermore, some aggregates of beta amyloid accumulate throughout brain tissue also in normal ageing. As it is cleaved and destroyed, more beta amyloid is formed and accumulates; the damage begins when the beta amyloid becomes concentrated in senile plaques and an inflammatory reaction with ongoing oxidative stress occurs. Axon of a neuron uses microtubules to transport substances between the center of neuron and its periphery. The assembly and structural integrity of microtubules are dependent upon several factors, the most important of which is a protein called 'Tau'. When Tau is abnormally phosphorylated, it forms the paired helical filaments known as neurofibrillary tangles. The similarity between aluminum induced tangles and AD pathology manifests only when the tissue is treated with silver stain and then viewed under light microscope (17). Detailed analysis showed that AD tangles are in fact; quite different from the pathology induced by aluminum. Aluminum induced tangles differ from those of AD in their distribution at both gross and ultra structural levels. While both types of tangles are found in the cortex and hippocampus, only aluminum induced pathology is also found in the spinal cord. Indeed with aluminum induced tangles, the spinal cord burden appears to exceed that of the brain itself. Within single neuron, aluminum induced tangles are found in the perikaryon and the proximal parts of the dendrites and axon. In contrast, AD tangles are found throughout the axons, including the terminals (Fig. 1 ).
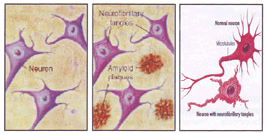
Fig 1: Amyloid Plaques and Neurofibrillary tangels in Normal and AD brain.
Aluminum mediated oxidative stress in brain
Aluminum is the trivalent cation that does not undergo redox changes. It has nonetheless been implicated in a variety of neurological disorders that have been associated with an increase in the formation of reactive oxygen species (ROS) such as hydrogen peroxide (H2O2) and the superoxide radical (O*-2). These species are generated by electron leakage from enzymes involved in the mitochondrial electron transport chain, which contain transition metal ions at their active sites (30). Although the exact mechanism of aluminum toxicity is not known, however, accumulating evidence suggests that the metal can potentiate oxidative and inflammatory events leading to tissue damage (31). The brain is particularly sensitive to oxidative stress due to increased level of oxidative damage and decrease level of antioxidants (32). Furthermore, neurons appear to be particularly vulnerable to free radicals as the important natural antioxidant glutathione content is low, they have higher membrane content of polyunsaturated fatty acids and brain requires substantial quantities of oxygen for metabolism. Moreover, many neurotransmitters are autooxidisable (Dopamine, Noradrenaline) and they react with oxygen to form superoxide, hydrogen peroxide and reactive quinines as well as Fe are found through out the brain and important Fe containing proteins in brain includes cytochromes, ferritin and tyrosine (21).
Mitochondria are one of the sensitive targets of oxidative stress in adult neurons (Wallace 1992). This may be due to the fact that mitochondrial DNA (mt DNA) does not encode for any repair enzymes and unlike nuclear DNA, it is not shielded by protective histones. Whereas mtDNA is in close proximity to the site where free radicals are generated during oxidative phosphorylation (33). Oxidative damage not only occurs to the proteins comprising the lesions of Alzheimer' disease, i.e., neurofibrillary tangles and senile plaques, but also precedes lesion formation in neurons at a risk of death during the disease. It has been reported that oxidative stress may not be the primary etiology of the disease, it precede specific cellular and tissue damage that underlies the onset of dementia (19). The direct evidence supporting increased oxidative stress in AD is an increased brain Fe, Al and Hg, capable of stimulating free radical generation (34–36).
Aluminum is considered one of the contributing factors to oxidative stress, as it generates ROS. Aluminum has been shown to cause oxidative damage to neurons through Iron (Fe2+) (37). It has been reported that aluminum stabilize ferrous (Fe2+) ion by reducing its rate of oxidation. Fe2+ promotes the generation of oxidative species, as it actively catalyzes the Fenton reaction (38). The Fenton reaction leads to formation of OH*, OH- and Fe3+ as aluminum suppress the superoxide dismutase (SOD) and catalase. Superoxide radical is readily converted to H202, and the breakdown of H2O2 to H2O and O2 by catalase is slowed down. Excess accumulation of H2O2 further leads to the production of OH* radicals, which in turn damages various proteins, DNA and membrane lipids (39,40).
Alteration in cell membrane structure and function in AD has been shown in several studies (41,42) and aluminum has also been demonstrated to modify biophysical property of the membrane structure and function. Aluminum is also reported to disturb the resting membrane potential, voltage activated ionic channels, transmitter's secretions as well as trans membrane potential differences and these alterations in cell membrane may be either due to direct interaction of aluminum with membrane proteins or induction of alterations in the lipid matrix (36). Aluminum appears to bind to the membrane polar heads resulting in injurious consequences for biological transport process and cellular metabolism (42).
Antioxidants in cognitive dysfunction
There are a number of reports implicating oxidative stress as one of the most important contributing factor in the development of AD. It has been reported that antioxidant supplements help to block the process of β-amyloid aggregation. According to free radical hypothesis of AD, numerous approaches for an effective antioxidants neuroprotection have been developed. Many free radical scavengers have been used in experimental paradigms of neuronal cell death in vitro and vivo. A number of experimental studies have shown that free radical scavenging substances inhibit the toxic effect of beta amyloid or superoxide on cell cultures and organs-typic hippocampus culture (43,44). Oxidative stress is reported to be involved in the early phase of Alzheimer's disease and therapeutic interventions then may be more useful. Commenges and Coworkers (45) found an inverse relationship between the intake of flavonoids and the risk of incident dementia of which AD is one possible cause. Furthermore, it has been reported that Vitamin E supplements may be associated with a reduced risk for AD (46). Also, Engelhart and coworkers (47) assessed the relationship between dietary intake of beta carotene, flavonoids , vitamin C and vitamin E and related risk of AD. Sharma and Gupta (48) suggested that alpha lipoic acid act as potent free radical scavenger and it helps in preventing cognitive impairment, oxidative stress, dementia, age and age related disorders such as AD, Parkinson. Large population based prospective COHART study results obtained after a mean follow up six years indicate that high dietary intake of vitamin C and vitamin E is associated with a significantly lower risk for AD (49) (Table no.2).
Involvement of Al in the generation of ROS:-
|
Al facilitate iron driven biological oxidation by formation of aluminum superoxide ion (AlO2*) and it act as a prooxidant by both catalyzing the formation of H2O2 and reducing Fe3+ to Fe2+. Fe2+ + O2 → Fe3+ + O2−. 2O2 + 2H+ → H2O2 + O2 2O2 − Al3+ → AlO2*2+ (2H+) → H2O2 + Al3+ and Fe3+ + A1O2*2+ → Fe 2+ + O2 there by facilitating the reaction Fe2+ + H2O2 OH* + OH− + Fe3+ Ferric and ferrous forms of iron can also react with superoxide anion and hydrogen peroxide to produce molecular oxygen and hydroxyl radical (OH*) respectively: Fe3+ + O−2 → Fe2 + O2 Fe2+ + H2O2 → Fe3+ + OH* + OH− (Fenton reaction) Hydroxy radical can be formed from superoxide anion and via Haber Weiss reaction :- O−* + H2O2 → O2 + OH− + OH* |
| ANTIOXIDANTS | PATEINTS WITH EARLY AD |
PATEINTS WITH MODERATE TO ADVANCE AD |
|---|---|---|
| Natural beta carotene | 30 mg/day | 30 mg/day |
| Vitamin E d-α Tocopherol succinate + d-α Tocopherol | 400IU/day | 600IU/day |
| Vitamin C (Calcium ascorbate) | 2g/day | 2g/day |
| Selenium | 250μg/day | 200μg/day |
| Co enzyme Q10 | 90mg/day | 120mg/day |
| NADH | 10mg/day | 20mg/day |
| N-Acetyl cysteine | 250mg/day | 1000mg/day |
| α-Lipoic acid | 30mg/day | 60mg/day |
Medical therapy:
Alzheimer's disease is a slowly progressing disease, starting with mild memory problems and ending with severe brain damage. The course disease takes and how fast changes occur vary from person to person. On average, AD patients live from 8 to 10 years after they are diagnosed, though the disease can last for as many as 20 years. Although prescription medications neither stop nor slowdown the progression of Alzheimer's dementia, Cholinesterase inhibitors such as tacrine, donepezil, matrifonate and rivestigmine might improve system in people with mild to moderate cases. Another drug mementine (Namenda) has been approved for treatment of moderate AD. Acetylcholine inhibitors (such as Tacrine, Donepezil, Metrifonate and Revastigmine) are currently prescribed after disease has actually developed. This class of drugs increases the amount of neurotransmitter, acetylcholine at the nerve terminal by decreasing its breakdown by the enzyme Cholinesterase. Acetylcholenesterase inhibitors have been shown to modestly improve memory and language and decrease the emotional symptoms of apathy, anxiety, hallucination, inappropriate behavior, and abnormal movement (51).
Conclusion
Healthy human body has effective barriers, such as skin, lungs and gastrointestinal tract against aluminum. Aluminum is not a nutrient, in other words the body has no need for this metal and avoidance has no negative consequence. The harmful forms of aluminum enter our foods as additives, such as emulsifier, acidifying agent, anticaking agent or coloring, cooking utensils etc. Packaging and handling food in aluminum containers increases the amount of Al in foods. Natural sources may be drinking water, vegetables and air. There are a number of ways for minimizing our exposure to aluminum. Al containing antidepressants can easily be avoided, as can aluminum utensils and even to play it safe, Al containing antacids. Commercially processed food such as pancake, wines, frozen dough and rising flours, are source of dietary Al. Their ingestion should be minimized. Sodium Aluminum phosphate an ingredient in baking powder, pickle and cheese should also be avoided. Besides minimizing aluminum exposure, taking the recommended dietary allowance (RDA) of calcium, magnesium, zinc and antioxidants should help to protect against Al accumulation (52).
Acknowledgement
We acknowledge with thanks the financial assistance from Indian National Science Academy, New Delhi to MH as Senior Scientist and from Indian Council of Medical Research (ICMR), New Delhi for Adhoc grant No.59/22/2004/BMS/TRM.
References
1. ATSDR C Ageuey for toxic substances and disease registry. Toxicological profile for aluminum US department of Health and Human Services. Public Health Service. 1990.
2. Savory j, Huang y, Herman MM, Reyes MR, wills MR Tau immunoreactivity tangles. Neuro Toxicology, 1997; 18: 63–67.
3. Siem, Quoted iu: Doellken, Ueber die Wirkung des aluminum besonderer Berucksicjtigung der durch das aluminum verusachten Lesion in centralnervensystem, Naunjyn-schmiedenbergs, Archive fur Experimentally Path and Parm. 1887; 40: 58–120.
4. McLachlan DRC, Bergeron C, Smith Boomer D, Rifat SL. Risk for neuropathologically confirmed Alzheimer's disease and residual aluminum in municipal drinking water employing weighted residential histories. Neurology 1996; 46: 401–5.
5. Klatzo I, Wisniewski HM Streicher E. Experimental production of neurofibrillary degeneration J neuropath & Exp Neurol. 1965; 24: 187–1999.
6. Wisniewski HM Terry RD Pena C, Stricher E, Klatzo I Experimental production of neuro fibrilacy degeneration J Neuropath of Exp Neurol 1965; 24: 139.
7. Terry RD Pena, C. Experimental production of neurofibrillary degeneration J. Neuropath of Exp Neurol 1965; 24: 200–210.
8. Berlyne GM, BenAri, J. Pest D, Weinberger J, Stern M, Gilmore GF, Levine R: Hyper aluminum aemia from aluminum resiness in renal failure. Lancet 2; 494: 1970.
9. Crapper DR, Dementia: Recent observations on Alzheimer's disease and Experimental aluminum encephalopathy in : Frontiers in neurology and neuroscience research p 97, edited by P Jeeman and GM Brown, University to Toronto Press, Toronto, 1974.
10. Crapper DR, Krishanans's Quittkat S: Aluminum neurofibrillary degeneration and Alzheimer's disease brain 99; 67: 1976.
11. Ebner K, Liss L, Couri D, Chou M: Relationship between the neurofibrillary tangles neuroaxonal degeneration, aluminum level and clinical symptomology. Federation proceedings 34, 3564a, 1975.
12. Trapp GA, Miner GD, Zimmerman RLMastri, AR, Heston LL, Aluminum levels in brain in Alzheimer's disease. Biol Psychiat 1978; 13: 709–718.
13. Yokel RA, Allen DD, Ackley DC. The distribution of A1 into and out of the brain. J In org Biochem 1999; 76: 127–132.
14. Perl DR good PF Uptake of A1 in to CNS along Nasal olfactory Pathways. Lancet 1987; I: 1028.
15. Roskamas AJ, Connor JR Aluminum access to the brain: a role for transferring and its receptor. Proc. Natl. Acad Sci. USA1990; 87: 9024–9027.
16. Gerhart DZ, Enerson BE, Zhdankina OY, Leino RL, Drenes LR Expression of monocorboxy transporter MCT 1 by brain endothelium and glia in adult smoking rats AM S Physiol 1997; 273: E207–213.
17. Wisniewski HM, Wen GY. Aluminum and Alzheimer's disease. Aluminum in biology and medicine. Wiley, Chichester (Ciba Foundation Symposium 169), 1992; pp. 142–164.
18. Flynn BL, Ranno AE. Phasmocolgic management of Alzheimer disease, Part II: Antioxidants anti hypertensives and ergoloid derivatives. Ann Pharmocother 1999; 33: 188–97.
19. Clegg A, Bryant J, Nicholson T, Mclutyre L, De Brose 's , Generd K, Wough N. Clinical and cost effectiveness of donepenzil, revastigmine and galautarnine for Alzheimer's disease: a rapid and systematic review Health Technol Assess. 2001; 5:1–137.
20. Bush TL, Miller SR, Criqui MN, et al Risk factors for morbidity and mortality in older population: are epidemiological approach in : Hazard WR, B Eirman EL, Blass JP et al, editors principles of Geriatric Medicines and gerontology, New York, McGraw Hill 1994; 153–166;.
21. Gupta YK, Sharma Monish, Chaudhary Geeta., Oxidative stress in Alzheimer's Disease and central Eschemia: Implication for antioxidant treatment (book review) 2004, Pharmacological perspectives of toxic chemicals and their antioxidants (24); 431–442.
22. Martyn CD, Ogmand C, Edwordson JA, Barker DJP, Harris EC, Lacey RE Geugraphical relation between Alzheimer disease and aluminum the drinking water Lancet 1989; 1: 59–62.
23. Flater TP Geographical associations between aluminum the drinking water and death rates with dementia perkinson's disease and amyotrophic lateral sclerosies in Norway. Trace Elem Med 1987; 4:179–80.
24. Neri LC, Hewitt D. Aluminum Alzheimer disease and drinking water. Lancet 1991; 338: 390.
25. Forbes WF, McAincy CA. Aluminum and dementia. Lancet 1992, 340: 668–9.
26. Campbell A, The potential role of aluminum in Alzheimer's disease. Nephrol Dial Transplant. 17: Suppl 2002; 2: 17–20.
27. Shimizu H, Mori T, Koyama M et al. Brain aluminum levels sharply increase in the elderly 'non-demented' as compared to younger adults. Nippon Ronen Igakkai Zasshi 1994; 31: 950–960.
28. Armstrong RA, Anderson J, Cowburn JD et al. Aluminum administered in drinking water but not in the diet influences biopterin metabolism in the rodent. Biochem Hoppe Seyler. 1992; 373: 1075–1078.
29. Evans P, Harrington C. Aluminumsilicate particulate and beta-amyloid in vitro interactions: a model of Alzheimer and beta- amyloid in vitro interactions: a model of Alzheimer plaque formation. Biochem Soc Trans 26: S251.
30. Hallwell B. Reactive oxygen species and the central nervous system. J Neurochem. 1992; 59: 1609–1623:.
31. Campbell A, Prasad KN, Bondy SC. Aluminum-induced oxidative events in cell lines: gloma are more responsible than neuroblostoma. Free Radic, Biol, 1999; 26 (9–10): 1166–16-71.
32. Boveris A, Chance B: The mitochondrial generation of hydrogen peroxide general properties and the effect of hyperbaric oxygen. Biochem Jl973; 134: 707–716.
33. Shoffner JM, Boown MD, Torroni., Lott MT, Cabell MF; Mirra SS, Beal MF, Yang CC, Gearing M, Salvo R, et al: Mitochondrial DNA varients observed in Alzheimer's disease and Parkinson's disease patients. Genomics 17: 171–184, 1993.
34. Markesbery WR. Oxidative stress hypothesis in Alzheimer's disease. Free Radic Biol Med 1997; 23:134–47.
35. Xu N, Majidi V, Markesbery WR, Ehman WD. Brain aluminum in Alzheimer's disease using an improved GFAAS method. Neurotox, 1992; 13: 735–744.
36. Gupta VB, Suram A, Hegde MI, Zecca L, Gamito RM., Ravid R, Shanker SK, Stein R, Shanmngavelu R, Jaganatha Rao KS,.Aluminum in Alzheimer's disease: we still at a cross road ? CMLS, Cell Mol Life Sci 2005; 62: 001–16.
37. Gutteridge JMC, Quinlan GJ, Clark I, Halliwell B. Aluminum salts accelerate peroxidation of membrane lipid stimulated by iron salts. Biochem Biohvs Acta 1985; 835: 441–447.
38. Ellen YY, Guo-Ross SS, Bondy SC. The stabilization of ferrous ion by a toxic beta amyloid fragment and by an aluminum salt. Brain Res 1999; 839: 221–226.
39. Bondy SC, Kritein S.. The promotion of iron induced generation of ROS in nerve tissue by aluminum. Mol Chem Neuropathoi 1996; 27: 185–194.
40. Ferretti G, Machionni I, Bacchetti T, Galezzi T, Dousset N. Effect of aluminum on lipid peroxidation of human high-density lipoproteins. Free Rad Res. 2003; 37: 515–521.
41. Zsiros V, Rojik I, Kovacs T, Csoti T, Erdely L. Comparative electrophysiological aspects of aluminum actions on cettral neurons and neuronal synapses of invertebrates and vertebrate animals. Neurotoxicology 1998; 19: 553-*556.
42. Weis C, Haung A. Aluminum altered membrane dynamics in human red blood cell white ghosts. Thrombosis Res. 1989; 54: 141–149.
43. Pappolla M.A, Sos M, Omar RA et al. Melatonin prevents death of neuroblastoma cells exposed to the Alzheimer amyloid peptide. J Neurosci 1997; 17: 1683–90.
44. Bastianetto S, Ramassamy C, Christen Y, Poirier J, Quirion R. Ginkgo biloba extract (EGb 761 ) prevents cell death induced by oxidative stress in hippocampal neuronal cell cultures. In: Packer L, Christen Y, Eds. Ginkgo biloba extract (EGb 761) study: lesson from cell biology. Advances in EGb 761 research 1998;. 7: 85–99.
45. Commenges D, Scotet V, Renaud S, et al Intake of flavonoids and oisk of dementia. Eur J Epidemio 2000; 16: 357–363.
46. Morris MC, Evans DA, Bienias JL et al. Dietary intake of antioxidant nutrients and the risk of incident of Alzheimer's disease in a biracial community study JAMA. 2002; 287: 3230–3237.
47. Engelhart MJ, Greenlings MI, Ruitenberg A, et al., Dietary intake of antioxidants and the risk of Alzheimer disease. JAMA. 2002; 287: 3223- 3229.
48. Sharma M, Gupta YK, Effect of alpha lipoic acid on intra cerebroventricular streptozotein model of cognitive impairment in rats. Eur Neuropsychopharmacol 2003; 13 (4): 241–7.
49. Morris MC, Beckett LA, Scherr PA. et al, Vitamin E and Vitamin C supplement use and risk of incedent Alzheimer's disease. Alzheimer's Dis Assoc Disord 1998; 12: 121–6.
50. Kedar N Prasad, Willium C Cole, K Prasad. Risk factor for Alzheimer's disease; Role of multiple Antioxidants. Non-steroidal anti inflammatory and cholenergic agents alone or in combination in prevention and treatment. J Ame coll Nut 2002; .21:, 506–522.
51. Ibach B, Haen E., Acetyl Cholinesterase inhibition in Alzheimer's disease. Current Pharmaceutical design 2004; 10: 231–251.
52. Foster HD Health, Disease and the environment 1992; 311–16.
(c) Annals of Neurosciences.All Rights Reserved