Annals of Neurosciences, Vol 14, No 4 (2007)
Annals of Neurosciences, Volume 12, Issue 4 (October), 2005
CYTOTOXIC EFFECTS OF 4-HYDROXYNONENAL IN PC 12 CELLS: INVOLVEMENT OF NEUROTRANSMITTER RECEPTORS
Corresponding Author:
Dr. A B Pant
Scientist,
In Vitro Toxicology Laboratory
Industrial Toxicology Research Centre,
P.O. Box: 80, MG Marg,
Lucknow-226001 (UP) India
Voice: +91–522–2627586 Ext.: 321 (Work)
+91 -9935044044 (Mobile)
FAX:+91–522–2628227
Email:
(Recieved on 26.09.2007)
Abstract
Effect of 4-hydroxynonenal (HNE) was studied on the sensitivity of various neurotransmitter receptors in PC-12 cells. Initially, cytotoxicity profiling of HNE was carried out using HNE concentrations 0.1–50mM for 30min to 24h. The endpoints selected were, trypan blue dye exclusion, MTT, LDH release and neutral red uptake (NRU) assays. Significant cytotoxic responses were observed by minimum 2h of exposure, except for HNE at 50mM, where cytotoxicity was exerted even at 90min. HNE concentrations 10–50mM were found to be cytotoxic, 2–5mM cytostatic and 0.1-lmM non-cytotoxic. Neurotransmitter receptors studies were carried out using specific radioligands with selected doses of HNE (1,10,25 and 50mM for l-8h). Significant decrease in binding of 3H-QNB, 3H-Fluinitrazepam and 3H-Ketanserin, known to label cholinergic-muscarinic, benzodiazepine and serotonin 5HT2A receptors respectively was observed even at lh exposure with 25 and 50mM concentrations of HNE. Whereas, significant inhibition in binding of 3H-Spiperone to DA-D2 receptor was started at 4h of exposure and continued till 8h. Specific binding with 3H-Fluinitrazepam and 3H-Ketanserin was reached to saturation at dose of 50mM for 4h and onwards. PC-12 cells have shown particular vulnerability to cytotoxic concentrations of HNE. Experimental HNE exposure provides an intriguing model of toxicant-cell interactions, which likely involved receptors in HNE neurotoxicity, lead neurodegeneration.
Key Words: Cytotoxicity, PC-12 cells, HNE, Neurotransmitter receptors
Introduction
Lipid peroxidation has been reported as one of the key factors in the etiology of variety of neurodegenerative diseases including cerebral ischemia/reperfusion. Alzheimer's disease and Parkinson's disease (1–3). Of the various lipid peroxidation products, 4-Hydroxy-trans-2-nonenal (HNE), a long-chain a, b-unsaturated aldehyde product is generated mainly by the oxidation of w-6 polyunsaturated fatty acids (4). At higher concentrations, HNE is reported to cause cytotoxicity and oxidative stress-induced cell death in many cell types including, PC-12 cells, HepG2 cell, V79 cell, cerebellar granule neurons, human osteosarcoma cells and fibroblasts and primary cultures of normal human osteoblasts (5–9), etc. Whereas, lower concentrations have also been reported to have modulatory effects on cell proliferation, inhibition in the synthesis of nucleic acids and proteins, stimulation of neutrophil Chemotaxis, stimulation of phospholipases, and activation of stress signaling pathways (1, 10–14). Induced levels of HNE are also been reported to cause neuronal cell death by oxidative stress mediated inhibition NF-kB signaling pathway (15).
As far as, the involvement of neurotransmitter is concern, have demonstrated the degeneration of dopaminergic neurons due to toxic responses by induced levels of HNE in pesticide challenged mice (16). Recently, it has also been suggested the possible association of significant inhibitions in the expression of receptor proteins with the induction of cytotoxicity and oxidative stress signaling pathways (17). The critical role of cytotoxic doses of HNE in the induction of neurotoxicity has been demonstrated in catecholaminergic neurons of ferritin transgenic mice (16). However, the effect of different neurotransmitters in HNE induced neurodegeneration has yet to be explored.
Thus, the present investigations were carried out to study the influences of cytotoxic doses of HNE on the expression of various neurotransmitter receptors in PC-12 cells. PC-12 cells were employed in the study, since they are most frequently used neuronal cells and have been found successful over the years to study neuronal functions due to the prominent expression of variety of neurotransmitter receptors in them (18–19).
Materials and Methods
Cell culture: PC-12 cell line, rat pheochromocytoma cells, used in all the experiments were originally procured from National Centre for Cell Sciences, Pune, India, since then it has been maintained at IN Vitro Toxicology Laboratory, Industrial Toxicology Research Centre, Lucknow, India. Cells were grown in nutrient mixture F-12 (Ham), 82.5% supplemented with 2.5% fetal bovine serum, 15% horse serum, 0.2% sodium bicarbonate and antibiotic and antimycotic solution (10X, 1ml/100ml of medium, In Vitrogen, Life technologies, USA). The cells were grown in 5% CO2 95% atmosphere in high humidity at 37°C. Each batch of cells was assessed for cell viability by trypan blue dye exclusion test, prior to select for experiment and batches showing more than 95% cell viability were used in the study. Cells of passage number between 18–25 were used in the study.
Reagents and consumables: All the specified chemicals, reagents, diagnostic kits, were purchased from Sigma Chemical Company Pvt. Ltd. St. Louis, MO, USA, unless otherwise stated. Nutrient mixture F-12 (Ham) culture medium, antibiotics, fetal bovine serum and horse serum were purchased from Gibco BRL, USA. Culture wares and other plastic wares used in the study were procured commercially from Nunc, USA. Milli Q water (double distilled deionized water) was used in all the experiments. Radioligands (3H-Spiperone, 3H-QNB, 3H-Fluinitrazepam and3H-Ketanserin) and competitors (Haloperidol, Atropine sulphate, Diazepam and Cinanserin) used in the receptor binding assay were procured commercially from NEN products, Boston, MA, USA.
HNE (4-Hydroxynonenal): 4-hydroxy trans-2-nonenal (HNE), an unsaturated aldehyde, proposed toxin was procured commercially from Cayman Chemicals, USA (Catalog No. 32100, CAS Registry No. 75899–68–2).
Experimental design: Cultured healthy cells were exposed to various concentrations (0.1–50mM) of HNE for a period of 30 min to 24hr. Following the respective exposures of HNE, cells were subjected to assess the cytotoxic responses with the help assays viz., trypan blue dye exclusion test, MTT, LDH and NRU assays. Further, experiments were carried out with selected doses of HNE (1,10,25 and 50mM for 1–8hr) to study the involvement, if any, of different neurotransmitter receptors viz., dopamine, cholinergic, benzodiazepine and serotonin-2A (5HT2A), in the metabolism/toxicity of HNE in cultured PC-12 cells.
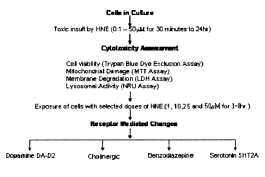
Cell Viability by Trypan Blue Dye Exclusion Test: The test was conducted to study the cell viability by assessing the loss of membrane integrity following the method of Pant et al. (2001) (20) with desired modifications. In brief, the cells (1x106) were seeded in T-25cm2 culture flasks (Nunc, Denmark) and allowed to grow for 24h in 5% CO2-95% atmosphere at 37°C under high humid conditions. Then the medium was replaced with serum free medium supplemented with either of concentration of HNE i.e., 0.1, 1, 2, 5, 10, 25 or 50μM. The treated cells were then allowed to incubate for intervals of 30min, 60min, 90min, 2h, 4h, 6h, 8h, 12h and 24h. Immediate after the completion of respective incubations, cells suspensions were aspirated centrifuged at 600rpm for 5min and washed twice with sterile PBS (pH 7.4), prior to stain with trypan blue dye. The cell suspension was then mixed with trypan blue dye (0.4% solution) at a ration of 1:5 (dye: cell suspension) and placed in heamocytometer. The counting for live (unstained transparent) and dead (blue stained) cells was made at 100X magnification in phase-contrast inverted microscope (Leica DMIL, Germany). The untreated sets were also run simultaneously under the identical conditions and served as control.
Mitochondrial activity by MTT assay: The MTT assay was done following the method pf Pandey et al. (2006) (21) with desired modifications. In brief, cells (1x104 per well) were seeded in poly-L-lysine pre-coated 96-well tissue culture plates and allowed to adhere for 24h in CO2 incubator at 37°C. The medium was then replaced with the serum free medium containing different concentrations (0.1/μM-50μM) of HNE. The cells were exposed to the test chemical i.e., HNE for various intervals as described earlier. Tetrazolium bromide salt (5mg/ml of stock in PBS) was added 10μl/well in 100/μl of cell suspension 4h prior to completion of incubation periods. At the end of incubation period, the reaction mixture was carefully taken out and 200ml of DMSO was added to each well by pipetting up & down several times unless the content gets homogenized. The plates were kept on rocker shaker for 10 min at room temperature and then read at 550nm using Multiwell microplate reader (Synergy HT, Bio-Tek, USA). Untreated sets run under the identical conditions were served as basal control; whereas cells treated with manganese 10-3 and 10-4M were used as positive control.
Lactate Dehydrogenase (LDH) release assay: LDH release is a method to measure the membrane integrity as a function of the amount of cytoplasmic LDH released into the medium. LDH assay was carried out using the readymade commercially available kit (LDH Assay Kit, Bio Vision, CA, USA) following the exposure of various concentrations of HNE (0.1–50μM) for a period of 2–24h. The assay was based on the measurement of activity of LDH released from damaged cells. In brief, following the treatment, plates were incubated as per the experimental schedule in CO2 incubator and centrifuged at 250xg for 4min. Then supernatant of each well was transferred to a fresh flat bottom 96-well culture plate and preceded further for enzymatic analysis as per the instructions given in the kit.
Neutral red uptake Assay: Neutral red uptake (NRU) assay was performed following the protocols of Borenfreund and Puerner (1984) with desired modifications (22). Briefly, cells were exposed to various concentrations of HNE (0.1–50μM) for a period of 2–24h. On the completion of respective incubation periods, the test solution was aspirated and cells were washed with PBS twice. Cells were then incubated for 3h in medium supplemented with neutral red (50μg/ml). Then medium was washed off rapidly with a solution containing 0.5% formaldehyde and 1% calcium chloride. The cells were then subjected to incubate further for 20min at 37°C in a mixture of acetic acid (1%) and ethanol (50%) to extract the dye. The plates were then read at 540nm using multiplate reader (Synergy HT, Bio-Tek, USA). The values were compared with control sets, run under identical conditions without the test compound.
Neurotransmitter Receptor studies: Effect of 4-hydroxynonenal (HNE) was studied on the sensitivity of various neurotransmitter receptors including, dopaminergic, cholinergic, benzodiazepine and serotonin 5HT2A in PC-12 cells. The experiments were conducted to study the concentration (1,10, 25 and 50mM) and time (for l-8h) dependent changes following the method of Seth et al. (2002) with desired modifications (23). Specific radioactive ligands and unlabelled competitors were used for specific receptor.
Radioligands: Radioligands of high specific activity were used for the estimation of different neurotransmitter receptors. 3H-Spiperone (1x109M), 3H-QNB (1x10-9M), 3H-Fluinitrazepam (0.5xl0-9M) and 3H-Ketanserin (1x109M) were used for the assay of Dopamine-D2 (DA-D2), Cholinergic, Benzodiazepine and Serotonin-2A (5HT2A) receptors respectively.
Displacers/Competitor: Specific displacers/competitor was used for the assay of different neurotransmitter receptor subtypes. Haloperidol (1x106M), Atropine sulphate (1x10-6M), Diazepam (1x10-6M) and Cinanserin (1x10-6M) were used for assay of Dopamine-D2 (DA-D2), Cholinergic, Benzodiazepine and Serotonin-A2 (5-HT2A), receptors respectively.
Membrane Preparation: Cells of both treated and control groups were harvested and centrifuged at 1000rpm for 10 min at 4°C. The cell pellets were then suspended in 5mM Tris HCl buffer (pH 7.4) and sonicated. The sonicated cell suspensions were then centrifuged at 20,000rpm for 20min at 4°C and the membrane pellets were suspended in 40mM tris HCl (pH 7.4) and were stored at -80°C till assay. Prior to storage, protein estimation of each sample was done as per the standard protocol.
Receptor binding assay: Receptor binding assay were carried out using Robotic Liquid Handling System (Multi Probe II EX, Packard, USA). In Brief, multiwell plates were arranged on the deck as mapped with winprep software. Sequential addition of the assay components was also programmed. Separate sets of triplicate were used for total binding and non-specific binding. For total binding, buffer (160ml), membrane (50ml) and radioligand (40ml) were added to each well. Whereas, a competitor (20ml) was added along with buffer (140ml), membrane (50ml) and radioligand (40ml) for non-specific binding assay. The reaction mixture was then incubated for 20 minutes at 37°C followed by filtration of the contents over manifold using vacuum. Plates were then washed with buffer twice and filtered in similar way. Scintillation cocktail (70ml) was added to each well and plates were left overnight and binding was counted per minute (cpm) using Top Count-NXT (Packard, USA). The cpm count of each well was utilized to calculate the percent change in the specific receptor following the exposure of various concentration of HNE.
Statistical analysis: The results are expressed as mean and standard error of means (SEM) for 6 wells of at least three experiments, as indicated in the figures. ANOVA was employed to detect differences between the treated and control groups, p <0.05 was taken to indicate significant differences.
Results
Cytotoxicity Assessment: Cytotoxicity assays were carried out in cultured PC-12 cells by exposing then to various concentrations (0.1- 50mM) of 4-hydroxynonenal (HNE) for different time periods (30min to 24h). The assays selected in the study were: trypan blue dye exclusion test, tetrazolium bromide salt (MTT) assay, neutral red uptake (NRU) assay and lactate dehydrogenase (LDH) release assay. The results of these assays are expressed in percent control. The control sets are the untreated batches of cells run simultaneously under identical conditions. The highlights of the results for cytotoxicity assessment are summarized in Fig. 1–4.
Trypan blue dye exclusion test: Data indicates that initial exposure up to 90 min could not cause statistically significant reduction in viable cell count in PC-12 cells except in case of HNE concentration 50mM at 90min (34±2.8 % of control). The concentrations 10,25 and 50mM of HNE were found to initiate statistically high significant loss in cell viability at 2h (34±2.2, 45±1.6 and 50±2.8%of control respectively) post exposure, which gradually increased in further exposure periods and reached to the maximum reduction by the end exposure period i.e., 24h (35±2.0, 60±4.8, 69±0.9% of control respectively). In one interesting finding, HNE concentrations 1,2 and 5mM were found to cause no statistically significant reduction in viable cell count in any time interval selected in the study, except for 2h i.e., 15±0.6, 20±0.9 and 23±2.2 % control respectively. Overall response of HNE in PC-12 cells, assessed trypan blue dye exclusion test was concentration and time dependent, where, concentrations 10,25 and 50mM were found to be cytotoxic post 2h and onward exposure periods and 0.1mM concentration could not show any influence on normal cell growth (Fig. 1).
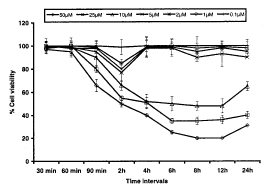
Fig.1. Assessment of percent cell viability in PC-12 cells following the exposure of various concentrations of HNE (trypan blue dye exclusion test). All values are mean±S.E.
Mitochondrial activity by MTT Assay: Highlights of the results of the MTT assay are summarized in Fig. 2. It is evident from the data that in the initial 90 min of exposure, only 50mM concentration of HNE was found to be the effective concentration where the reduction in percent cell viability could reach to statistically significant level (40±3..6% of control); Surprisingly, exposure period of 2h was found to be most effective for all the concentrations of HNE used in the study except 0.1mM. At this exposure period, a concentration dependent statistically highly significant (p<0.001) reduction in percent cell viability in PC-12 cells could be recoded up to minimum concentration of 1mM (21 ±0.9% of control). In general, HNE concentrations 10, 25 and 50mM have shown statistically highly significant loss in percent cell viability (38±2.6, 49±1.8 and 55±3.4% of control respectively) when exposed for 2h and above period and this reduction was time and concentration dependent too (Fig. 2). PC-12 cells were found tolerant and had not shown significant reduction in percent cell viability under the influence of 0.1 -5mM concentrations of HNE for the exposure period between 4–24h (Fig. 2). Although, overall trend of reduction in viable cell count was similar in both trypan blue dye exclusion test and MTT assay, but MTT assay was found to be more sensitivity then the trypan blue dye exclusion test (Fig. 1–2).
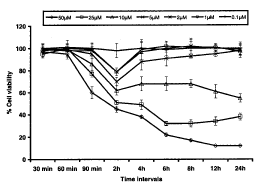
Fig.2. Assessment of percent cell viability in PC-12 cells following the exposure of various concentrations of HNE (MTT assay). All values are mean±S.E.
LDH release assay: Highlights of the results of the LDH release assay are summarized in Fig. 3. For LDH release assay, cells were exposed to various concentrations (0.1 – 50mM) of HNE for a period between 2 to 24h. Although, the concentration dependent statistically significant (p<0.001) increases in the LDH release was initiated even at 2h of HNE exposure, but this increase brought to the highest level by 4h of exposure, followed by a gradual decrease till the end of exposure period i.e., 24h. At all the time interval, increase in the LDH release levels were concentration dependent and HNE concentrations 2–50mM were found to pose a statistically significant effects on LDH release throughout the exposure period. Unlike the trypan blue dye exclusion test and MTT assay, HNE concentration 1mM could induce the levels of LDH release significantly between 4–8h of exposure. Whereas, like earlier tests, HNE concentration 0.1mM did not find effective at any time interval. In totality, more pronounced induction in the levels of LDH release could be recorded through out, even at lower concentrations of HNE (up to 1mM) in PC-12 cells under experimental conditions (Fig. 3).
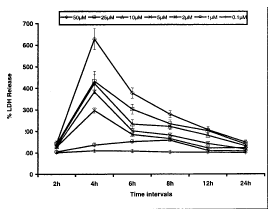
Fig.3. Assessment of percent change in LDH release in PC-12 cells following the exposure of various concentrations of HNE (LDH assay). All values are mean±S.E.
Neutral Red Uptake Assay: Data highlights of neutral red uptake (NRU) assay are summarized in Fig. 4. The assay was carried out by exposing PC-12 cells to various concentrations (0.1 – 50mM) of HNE for a period between 2 to 24h. Similar trend in reduction of percent cell viability like MTT assay was observed; however the level of reduction was comparatively less. HNE concentrations 10–50mM were found statistically significant at for all the exposure periods. At 2h of exposure, gradual and statistically significant (p<0.001 and p<0.01) decrease in percent cell viability could be recorded for HNE concentrations of 1–50mM. It is well apparent from the data that effect of HNE concentrations 1–5mM was insignificant in all the time intervals except 2h. HNE concentration 0.1mM was found universally ineffective to cause any adverse effect. Overall, this test system was found to be least sensitive than all other test methods adopted for the assessment of cytotoxicity of HNE in PC-12 cells under our experimental conditions (Fig. 1–4).
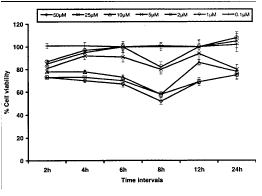
Fig.4. Assessment of percent cell viability in PC-12 cells following the exposure of various concentrations of HNE (NRU assay). All values are mean±S.E.
Neurotransmitter receptor studies: Result highlights of the experiments carried out to study the involvement, if any, of different neurotransmitter receptors viz., dopamine, cholinergic, benzodiazepine and serotonin-2A (5HT2A), in the metabolism/toxicity in PC-12 cells following the exposure of selected doses of HNE (1, 10, 25 and 50mM for 1, 4 and 8h) are presented in Fig.5–8.
HNE exposure and Dopamine-D2 (DA-D2) receptor: A dose dependent decrease in specific binding of 3H-Spiperone was observed. For 50mM of HNE concentration, statistically significant decrease (11 ±0.4% of control) could be recorded even at 1h incubation period, which further brought down to subsequent incubations i.e., 27±1.1% at 4h and 35±2.1% at 8h. HNE concentrations 10 and 25mM were also found to cause significant reduction in DA-D2 receptor binding at 8h of incubation period, whereas 1mM concentration was found ineffective in general for this receptor (2±0.1,2±0.1 and 4±0.2% of control at 1,4 and 8h respectively) (Fig.5).
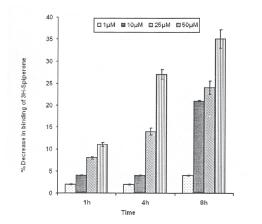
Fig.5. Percent inhibition in specific binding of 3H-Spiperone with DA-D2 receptor in the membrane harvested from PC-12 cells following the exposure of HNE. All values are mean±SE. (*p<0.01, **p<0.001 Vs Control)
HNE exposure and cholinergic receptors: Time schedule and doses of HNE treatment in PC-12 cells for cholinergic receptor binding were similar to DA-D2. Decrease in specific binding of 3H-QNB was statistically significant for all the concentrations of HNE used in the study at 1 and 4h exposure periods except for HNE 1mM after 1h exposure (14±0.8% of control). Though the trend of decrease in specific binding was similar to DA-D2, but the arithmetic values were quite high in case of 3H-QNB binding than 3H-Spiperone for all the concentrations of HNE used in both 1 and 4h incubation time points. However, unlike DA-D2, saturation was observed for 3H-QNB binding by 8h of exposure of 1mM of HNE. While rest of the concentrations of HNE (10, 25 and 50mM) were able reduce the 3H-QNB binding (39 ±2.1, 41 ±2.3,42 ±2.4% of control respectively) following 8h of exposure (Fig.6).
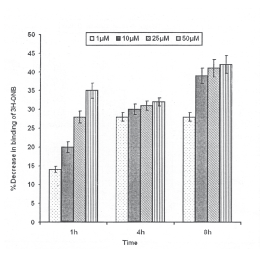
Fig.6. Percent inhibition in specific binding of 3H-QNB with cholinergic receptor in the membrane harvested from PC-12 cells following the exposure of HNE. All values are mean±SE. (*p<0.01, **p<0.001 Vs Control)
HNE exposure and benzodiazepine receptors: It is evident from the data that HNE concentrations 1 and 10mM could not cause a significant reduction in the specific binding of 3H-Flunitrazepam after 1h of exposure, where after the values for 10mM could reach to statistically significant level i.e., 38±2.3 and 45±2.3% of control respectively (p<0.01) at 4 and 8h of incubations. Whereas, in case of 1mM HNE, values were found to be saturated (15±1.0 and 16±1.1% reduction of control) at 4 and 8h of incubations. HNE 25mM concentration was found to cause statistically significant gradual decrease in the specific binding through out the incubations i.e., 1, 4 and 8h (20±1.1, 39 ±2.4 and 49 ±2.5% reduction of control respectively). However, 50mM HNE brought down the specific binding 46±2.3% of control with in 1h of exposure but, it was the saturation point, since in subsequent incubations (4 and 8h), there was no further decrease in the specific binding of 3H-Flunitrazepam (50±3.1 and 51 ±3.2% reduction of control) (Fig.7).
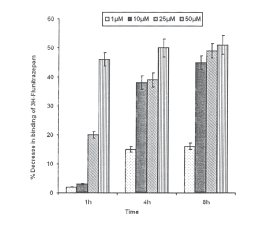
Fig.7. Percent inhibition in specific binding of 3H-Flunitrazepam with benzodiazepine receptor in the membrane harvested from PC-12 cells following the exposure of HNE. All values are mean±S.E. (*p<0.01, **p<0.001 Vs Control)
HNE exposure and serotonin 5HT-2A receptors: Statistically significant reduction in the specific binding of 3H-Ketanserin was observed even at 1h exposure of 25 and 50mM concentrations of HNE (20±0.6 and 41 ±1.4% reduction of control respectively) which decreased in subsequent incubation of 4h following the exposure of 25mM HNE only (31 ± 1.2% reduction of control), whereas saturation was observed for exposure of 50mM HNE (42±2.8% reduction of control). At 4h of exposure, HNE concentrations 1 and 10mM were also able to influence significant reduction in specific binding of 3H-Ketanserin (28±0.1 and 30± 1.3% reduction of control respectively). In general, saturation for specific binding was observed for all the doses of HNE used in the study by 8h of incubation, since no further decrease in specific binding could be recorded (Fig.8).
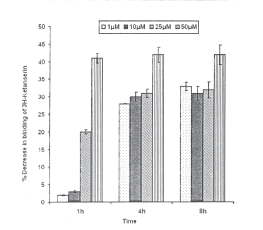
Fig.8. Percent inhibition in specific binding of 3H-Ketanserin with serotonin 5HT2A receptor in the membrane harvested from PC-12 cells following the exposure of HNE. All values are mean±S.E. (*p<0.01, **p<0.001 Vs Control)
Discussion
Cytotoxicity studies: In the present study, it has also been observed that HNE (10–50mM) exerted statistically significant physiological stress, which finally leads to cell death in PC-12 cells between 2–24h. In general, HNE concentrations 1–5mM was found to induce maximum damaging effects at 2h of exposure followed by sequential decrease in the response. This might be because of metabolism of HNE into non-toxic or low toxic metabolites in later time points. Since, the degradation of HNE into non-toxic/less toxic components under both intracellular and extracellular conditions have already been demonstrated (24–25). Higher concentrations of HNE (10–50mM) used in the study were found to cause significant increase in the detrimental effects with the increase in concentrations and exposure period. These finding can be suggestive the classical nature of HNE to exert the cytotoxicity by inducing signaling pathways for the production of cytokines, which mediates injury and apoptosis (26). The possible involvement of oxidative stress either by generating reactive oxygen species and reactive nitrogen species or by the activation of stress response transcription factors such as NF-kappa B, AP-1 and P53 can not be ignored (27–28). Investigations were carried out using four test systems to assess the cytotoxicity of HNE in PC-12 cells. LDH release assay was found to be the most sensitive, followed by MTT, trypan blue dye exclusion and NRU assay respectively. Cytosolic and mitochondrial catabolism is involved in LDH and MTT assays respectively, which support the reactive nature of HNE and its metabolites either directly to the proteins or oxidative stress signaling pathways carrying out there (29,30).
HNE concentration O.1mM was found at no effect level at all the time points in all the tests conducted for cytotoxicity. This correlates well with the earlier findings demonstrating the normal physiological concentrations of HNE in the plasma and erythrocytes of human and experimental animals between 1–10mM (31). In general, the HNE concentrations 10–50mM were found to be cytotoxic, whereas concentrations 2–5mM were cytostatic and 0.1–1mM was found to be non-cytotoxic in PC-12 cells, under our experimental setup. Existing literature using a variety of cell system including PC-12 cells HepG2, cell V79, cell cerebellar granule, neurons human osteosarcoma cells, and fibroblasts and primary cultures of normal human osteoblasts etc., have almost similar trends for cytotoxic responses for HNE (5–9).
Neurotransmitter receptor studies: The other objective was to study the involvement of different neurotransmitter receptors in the metabolism/toxicity of HNE in PC-12 cells. Cells were exposed to HNE concentrations 10–50mM (cytotoxic) and 1mM (non-cytotoxic) for the period of 1–8h. PC-12 cells were employed in the study, since they are most frequently used neuronal cells derived from sympathetic nerve pheochromocytoma and have been found successful over the years to study neuronal functions due to the prominent expression of variety of neurotransmitter receptors in them (18–19,32–33).
In general, cytotoxic concentrations of HNE 25 and 50mM were found to cause significant inhibition in the expression of receptor proteins of all the four receptors studied, even at 1h of exposure. The finding depicted the physiological functionality of the system and supporting to the finding that higher concentrations of HNE play a key role in the degradation of dopaminergic neurons by inducing various signaling pathways under clinical cases of Parkinson's diseases (PD), chemically induced in vivo and in vitro models of PD (3,16,34). Degeneration of dopaminergic neurons due to toxic responses by induced levels of HNE in pesticide challenged mice has also been reported recently. Since the cytotoxic response of HNE is directly or indirectly found to be associated with the oxidative stress mediated damages in variety of cells (6,8,9) thus, the possible association of significant inhibitions in the expression of receptor proteins with the induction of cytotoxicity and oxidative stress signaling pathways can not be ignored (6,8,9,17). To the best of our knowledge, we are reporting first time the inhibitory responses of cholinergic, benzodiazepine, serotonin 5HT2A receptors in PC-12 cells following the exposure of cytotoxic concentrations of HNE. Thus, the common pathways for degeneration of these receptor proteins are also anticipated through induction of cytotoxicity and oxidative stress signaling (35–36).
Increased exposure periods (4 and 8h) would have been expected to cause a progressive pattern of inhibition in the specific receptor binding. Instead, even after a continuous exposure of 8h, only the exposure of HNE for 1h with cytotoxic concentrations resulted in a significant reduction in specific binding to all four receptors studied. Thereafter, saturation in the receptor binding was observed for all the receptors except DA-D2. At least two interpretations may explain this finding. HNE neurotoxicity could preferentially affect receptors population in the cells and most of which are inhibited after 1h of exposure of HNE. On the other hand, it is also possible that, after 1h of exposure, defense mechanism in the cells allows them to resist for longer exposure periods i.e., 4 or 8h. Taken together, these results reveal a new paradigm of degradation of neurotransmitter receptors involved in various neurodegenerative disorders, in which neuronal fate appears to be strictly dependent upon the sequence of toxic challenges and the interplay between cell vulnerability, damaging effects and protective responses (16,37–38). However, continuous inhibitory responses of cytotoxic concentrations of HNE with increasing exposure period for DA-D2 receptor can be suggested on the bases of binding rate of reaction, since the arithmetic values of highest level of inhibition following 8h of exposure even could not cross to the level of inhibition at 1 h in any other receptor studied in the present investigations. Thus, there is a strong possibility that the receptors were available for specific binding till late exposure i.e., 8h.
No significant inhibitory response could be recorded except for cholinergic receptor, following the exposure of non-cytotoxic concentrations of HNE (1–10mM) for a period of 1h. Inhibition in the receptor proteins at lower concentrations began after 1h exposure and progressed over the following exposures i.e., 4 and 8h. This finding suggests the possibility that cumulative doses may play an important role for the development of response, specifically when cells are exposed to lower concentrations (1). However, we strongly thought that the short half-life of HNE under in vitro conditions using purified population of single cell type, the experimental exposure of HNE (1,4 and 8h) is unlikely to result in any significant accumulation of toxicant (31). An alternative explanation to these results may be the initial exposure (1h) acts as a 'priming' event, enhancing the cell vulnerability to a subsequent toxic insult (4 and 8h).
For benzodiazepine and serotonin 5HT2A receptors, saturation stages were observed even after 4h and 8h exposure of cytotoxic concentrations of HNE. This finding suggests that with the progression of continuous exposure of cytotoxic concentrations of HNE, the antioxidant signaling pathways could have induced in the cells to develop them relatively more resistance to oxidative injury (39). This can also be explained with the “priming effect hypothesis”, where although reversible, but initial injury may render neurons more vulnerable towards the oxidative challenges (12). Several lines of experimental evidence are available to further support a direct relationship between cytotoxicity and receptor degeneration in cells having specific neurotransmitter receptors. In one of experimental study using catecholaminergic neurons of ferritin transgenic mice, the critical role of cytotoxic doses of HNE in the induction of neurotoxicity has been demonstrated (16).
In summary, data of our study support the conclusion that PC-12 cells have shown the particular vulnerability to cytotoxic concentrations*of HNE. Experimental HNE exposure provides an intriguing model of toxicant-cell interactions in which, depending upon the exposure regimen and concentration of HNE, cytotoxicity mediated oxidative stress may be reversible or lead to neurodegeneration. The likely involvement of cytotoxicity signaling pathways in HNE neurotoxicity is also noteworthy. Besides HNE, a variety of metabolites of HNE may perhaps share the ability to cytotoxic pathways. This ability could therefore, characterize an entire class of lipid derived aldehyde neurotoxicants of particular interest for neurodegenerative processes by involving different receptors.
Acknowledgment:
The authors are grateful to Director, Industrial Toxicology Research Centre, Lucknow, for his keen interest in the present work. This is a part of the project work financially supported by Indian Council of Medical Research, New Delhi. The technical laboratory assistance by Mr. Rajesh Mishra is also acknowledged.
References:
1. Zhang Y, Milatovic D, Aschner M, Feustei PJ, Kimelberg HK. Neuroprotection by tamoxifen in focal cerebral ischemia is not mediated by an agonist action at estrogen receptors but is associated with antioxidant activity. Exp Neurol 2007; 204: 819–27.
2. Dmitriev LF. The involvement of lipid radical cycles and the adenine nucleotide translocator in neurodegenerative diseases. J Alzheimers Dis 2007; 11: 183–90.
3. Liang LP, Huang J, Fulton R, Day BJ, Patel M. An orally active catalytic metalloporphyrin protects against 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine neurotoxicity in vivo. J Neurosci 2007; 27: 4326–33.
4. Sakai T, Shimizu Y, Kawahara S. Effect of NaCl on the lipid peroxidation-derived aldehyde, 4-hydroxy-2-nonenal, formation in boiled pork. Biosci Biotechnol Biochem 2006; 70: 815–20.
5. Piga R, Saito Y, Yoshida Y, Niki E. Cytotoxic effects of various stressors on PC12 cells: involvement of oxidative stress and effect of antioxidants. Neurotoxicology 2007; 28: 67–75.
6. Gallagher EP Huisden CM, Gardner JL. Transfection of HepG2 cells with hGSTA4 provides protection against 4-hydroxynonenal-mediated oxidative injury. Toxicol In Vitro (online available) 2007.
7. Li D, Hinshelwood A, Gardner R, McGarvie G, Ellis EM. Mouse aldo-keto reductase AKR7A5 protects V79 cells against 4-hydroxynonenal-induced apoptosis. Toxicology 2006; 226: 172–80.
8. Arakawa M, Ishimura A, Arai Y, etal. N-Acetylcysteine and ebselen butnot nifedipine protected cerebellar granule neurons against 4-hydroxynonenal-induced neuronal death. Neurosci Res 2007; 57: 220–29.
9. Borovic S, Cipak A, Meinitzer A, etal. Differential sensitivity to 4-hydroxynonenal for normal and malignant mesenchymal cells. Redox Rep 2007; 12: 50–54.
10. Uchida K, Stadtman ER. Selective cleavage of thioether linkage in proteins modified with 4-hydroxynonenal. Proc Natl Acad Sci USA 1992; 89: 5611–15.
11. Wonisch W, Kohlwein SD, Schaur J, etal. Treatment of the budding yeast Saccharomyces cerevisiae with the lipid peroxidation product 4-HNE provokes a temporary cell cycle arrest in G1 phase. Free Radie Biol Med 1998; 25: 682–87.
12. Yu L, Ran PX. The expression of 4-hydroxy-2-nonenal caused by cigarette smoke condensate exposure in human bronchial epithelial cells and the interventional effects of ginkgolid B. Zhonghua Jie He He Hu Xi Za Zhi 2006; 29: 247–51.
13. Natarajan V, Scribner WM, Vepa S. Phosphatase inhibitors potentiate 4-hydroxynonenal-induced phospholipase D activation in vascular endothelial cells. Am J Respir Cell Mol Biol 1997; 17: 251–59.
14. Chen ZH, Saito Y, Yoshida Y, etal. 4-Hydroxynonenal induces adaptive response and enhances PC12 cell tolerance primarily through induction, of thioredoxin reductase 1 via activation of Nrf2. J Biol Chem 2005; 280: 41921–27.
15. Camandola S, Poli G, Mattson MR The lipid peroxidation product 4-hydroxy-2, 3-nonenal inhibits constitutive and inducible activity of nuclear factor kappaB in neurons. Brain Res Mol Brain Res 2000; 85: 53–60.
16. McCormack AL, Atienza JG, Johnston LC, atal. Role of oxidative stress in paraquat-induced dopaminergic cell degeneration. J Neurochem 2005; 93: 1030–37.
17. Abdul HM, Butterfield DA. Involvement of PI3K/PKG/ERK1/2 signaling pathways in cortical neurons to trigger protection by co-treatment of acetyl-L-carnitine and alpha-lipoic acid against HNE-mediated oxidative stress and neurotoxicity: implications for Alzheimer's disease. Free Radie Biol Med 2007; 42: 371–84.
18. Sutton LP, Honardoust D, Mouyal J, Rajakumar N, Rushlow WJ. Activation of the canonical Wnt pathway by the antipsychotics haloperidol and clozapine involves dishevelled-3. J Neurochem 2007; 102:153–69.
19. Dickinson JA, Hanrort KE, Mok MH, Kew JN, Wonnacott S. Differential coupling of alpha7 and non-alpha7 nicotinic acetylcholine receptors to calcium-induced calcium release and voltage-operated calcium channels in PC12 cells. J Neurochem 2007; 100: 1089–96 .
20. Pant AB, Agarwal AK, Sharma VP, Seth PK. In vitro cytotoxicity evaluation of plastic biomedical devices. Hum Exp Toxicol 2001; 20: 412–17.
21. Pandey MK, Pant AB, Das M. In vitro cytotoxicity of polycyclic aromatic hydrocarbon residues arising through repeated fish fried oil in human hepatoma Hep G2 cell line. Toxicol In Vitro 2006; 20: 308–16.
22. Borenfreund E and Puemer JA. A simple quantitative procedure using monolayer cultures for cytotoxicity assays. J tissue cult Meth 1984; 9: 7–9.
23. Seth K, Agrawal AK, Date I, Seth PK. The role of dopamine in manganese-induced oxidative injury in rat pheochromocytoma cells. Hum Exp Toxicol 2002; 21: 165–70.
24. Finkelstein EI, Ruben J, Koot CW, Hristova M, Van Der Vliet A. Regulation of constitutive neutrophil apoptosis by the alpha,beta-unsaturated aldehydes acrolein and 4-hydroxynonenal. Am J Physiol Lung Cell Mol Physiol 2005; 289: 1019–28.
25. Duarte AI, Santos MS, Oliveira CR, Rego AC. Insulin neuroprotection against oxidative stress in cortical neurons—involvement of uric acid and glutathione antioxidant defenses. Free Radic Biol Med 2005; 39: 876–89.
26. Shi Q, Vaillancourt F, Cote V, atal. Alterations of metabolic activity in human osteoarthritic osteoblasts by lipid peroxidation end product 4-hydroxynonenal. Arthritis Res Ther 2006; 8: 159.
27. Buzard GS, Kasprzak KS. Possible roles of nitric oxide and redox cell signaling in metal-induced toxicity and carcinogenesis: a review. J Environ Pathol Toxicol Oncol 2000; 19: 179–99.
28. Chung FL, Pan J, Choudhury S, atal. Formation of trans-4-hydroxy-2-nonenal- and other enal-derived cyclic DNA adduets from omega-3 and omega-6 polyunsaturated fatty acids and their roles in DNA repair and human p53 gene mutation. Mutat Res 2003; 531: 25–36.
29. Choudhary S, Xiao T, Srivastava S, atal. Toxicity and detoxification of lipid-derived aldehydes in cultured retinal pigmented epithelial cells. Toxicol Appl Pharmacol 2004; 204: 122–34.
30. Dwivedi S, Sharma A, Patrick B, Sharma R, Awasthi YC. Role of 4-hydroxynonenal and its metabolites in signaling. Redox Rep 2005; 12: 4–10.
31. Srivastava S, Dixit BL, Cai J, atal. Metabolism of lipid peroxidation product, 4-hydroxynonenal (HNE) in rat erythrocytes: Role of aldose reductase. Free Radic Biol Med 2000; 29: 642–51
32. Rosenström U, Sköld ?, Plouffe B, atal. New selective AT2 receptor ligands encompassing a gamma-turn mimetic replacing the amino acid residues 4–5 of angiotensin II act as agonists. J Med Chem 2005; 48: 4009–24
33. Homma K, Kitamura Y, Ogawa H, Oka K. Serotonin induces the increase in intracellular Ca2+ that enhances neurite outgrowth in PC 12 cells via activation of 5-HT3 receptors and voltage-gated calcium channels. J Neurosci Res 2005; 84: 316–25
34. Chen ZH, Yoshida Y, Saito Y, Noguchi N, Niki E,. Adaptive response induced by lipid peroxidation products in cell cultures. FEBS Lett 2006; 580: 479–83
35. Joseph JA, Fisher DR, Carey AN, Bielinski, DF. Dopamine-induced stress signaling in COS-7 cells transfected with selectively vulnerable muscarinic receptor subtypes is partially mediated via the i3 loop and antagonized by blueberry extract. J Alzheimers Dis 2006; 10: 423–37
36. Row BW, Kheirandish L, Cheng Y, Rowell PP, Gozal D. Impaired spatial working memory and altered choline acetyltransferase (CHAT) immunoreactivity and nicotinic receptor binding in rats exposed to intermittent hypoxia during sleep. Behav Brain Res 2007; 177: 308–14
37. Le W-D, Colon LV, Xie W-J, atal. Cell death induced by β-amyloid 1–40 in MES 23.5 hybrid clone: the role of nitric oxide and NMDA-gated channel activation leading to apoptosis. Brain Res 1995; 686: 49–60
38. Mark RJ, LoveU MA, Markesbery WR, Uchida K, Mattson MR A role for 4- hydroxynonenal in disruption of ion homeostasis and neuronal death induced by β-peptide. J Neurochem 1997; 68: 255–64
39. Kruman I, Bruce-Keller AJ, Bredsen D, Waeg G, Mattson P Evidence that 4-Hydroxynonenal mediates oxidative stress-induced neuronal apoptosis. J Neurosci 1997; 17: 5089–100.
(c) Annals of Neurosciences.All Rights Reserved